Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Moving to the next question prevents changes to this answer Question 29 Antudent mixos 40g of KNO3 with 10.0 ml. of water room temperature, and
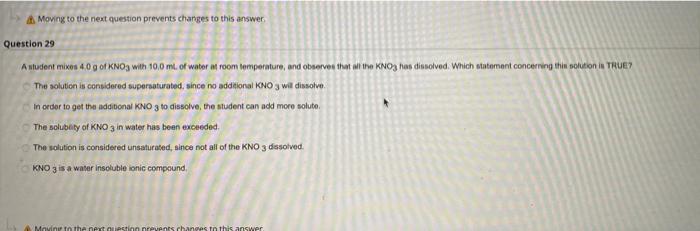 Moving to the next question prevents changes to this answer Question 29 Antudent mixos 40g of KNO3 with 10.0 ml. of water room temperature, and observes that at the KNO3 han disolved. Which statement concerning thin solution in TRUE? The solution is considered supersaturated, since no additional KNO3 wil dissolve. In order to get the additional KNO3 to dissolve, the student can add more soluto The solubaty of KNO3 in water has been exceeded The solution is considered unsaturated, since not all of the KNO3 dissolved. KNO3 is a water insoluble ionic compound Movin is
Moving to the next question prevents changes to this answer Question 29 Antudent mixos 40g of KNO3 with 10.0 ml. of water room temperature, and observes that at the KNO3 han disolved. Which statement concerning thin solution in TRUE? The solution is considered supersaturated, since no additional KNO3 wil dissolve. In order to get the additional KNO3 to dissolve, the student can add more soluto The solubaty of KNO3 in water has been exceeded The solution is considered unsaturated, since not all of the KNO3 dissolved. KNO3 is a water insoluble ionic compound Movin is

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


