Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Need help with these 3 problems pls helps Compare the following: Acid 1: phosphoric acid, H3PO4 Acid 2: hydrogen sulfide ion, HS Acid 3: carbonic
Need help with these 3 problems pls helps 
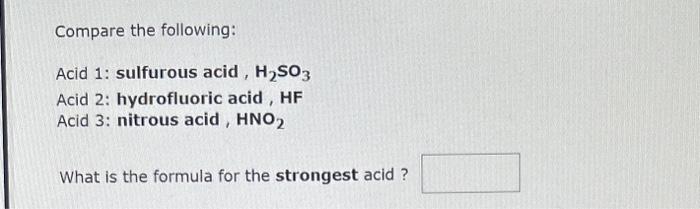
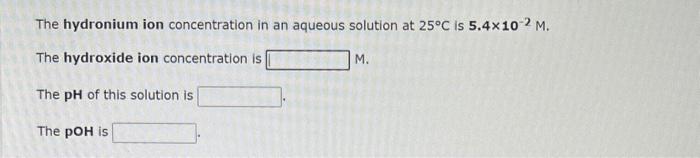
Compare the following: Acid 1: phosphoric acid, H3PO4 Acid 2: hydrogen sulfide ion, HS Acid 3: carbonic acid, H2CO3 What is the formula for the strongest acid? Compare the following: Acid 1: sulfurous acid, H2SO3 Acid 2: hydrofluoric acid, HF Acid 3: nitrous acid, HNO2 What is the formula for the strongest acid ? The hydronium ion concentration in an aqueous solution at 25C is 5.4102M. The hydroxide ion concentration is M. The pH of this solution is The pOH is 


Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


