Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Now calculate the specific enthalpies of the vapor ( V ) and liquid ( L ) streams of the evaporator with respect to the state
Now calculate the specific enthalpies of the vapor V and liquid L streams of the evaporator with respect to the state of the feed to the evaporator using the following data for the properties of the evaporator contents.
Then use those specific enthalpies to recalculate the mass flow rate of the required steam.
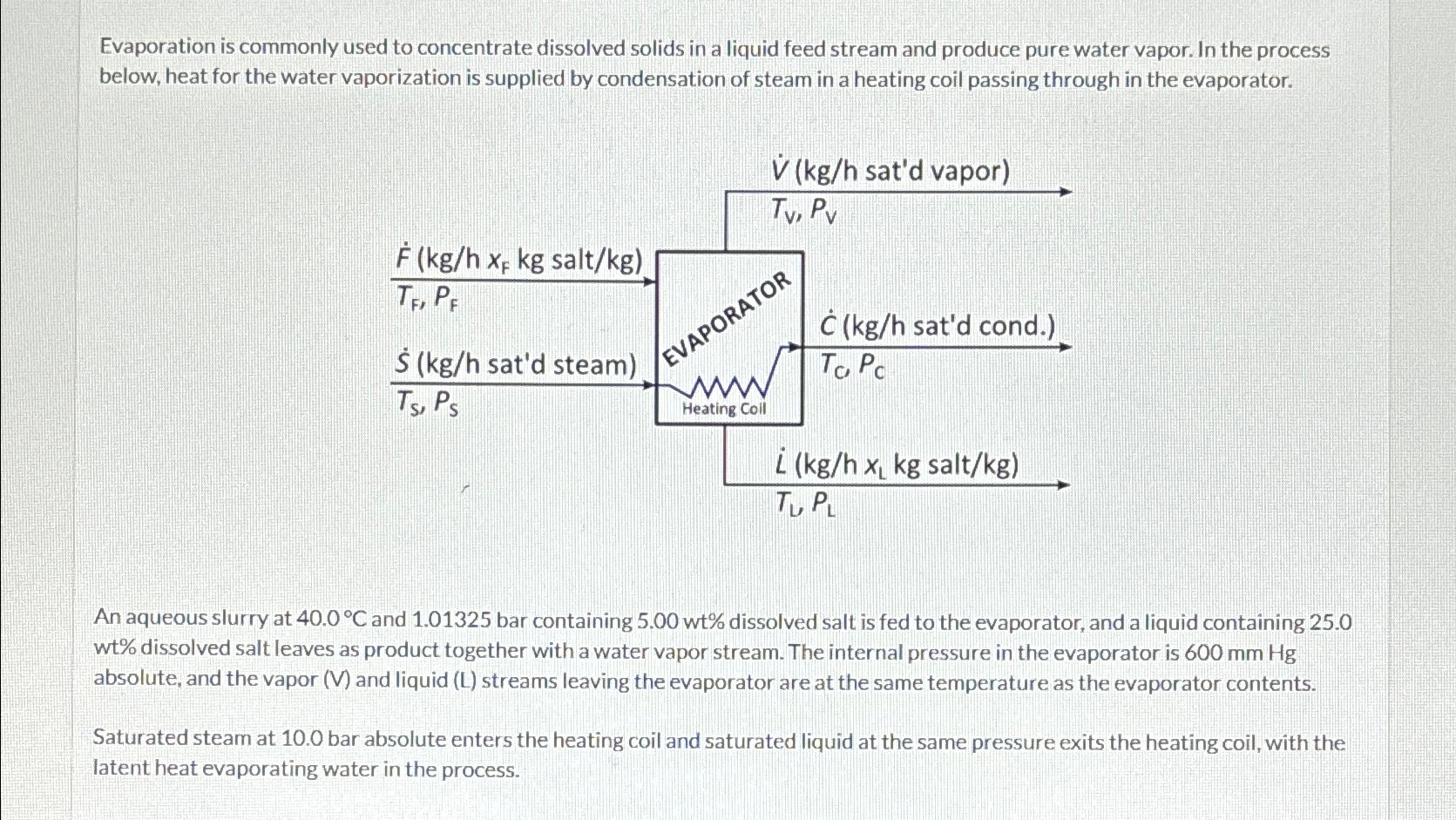
Evaporation is commonly used to concentrate dissolved solids in a liquid feed stream and produce pure water vapor. In the process below, heat for the water vaporization is supplied by condensation of steam in a heating coil passing through in the evaporator.
An aqueous slurry at and bar containing dissolved salt is fed to the evaporator, and a liquid containing wt dissolved salt leaves as product together with a water vapor stream. The internal pressure in the evaporator is absolute and the vapor V and liquid L streams leaving the evaporator are at the same temperature as the evaporator contents.
Saturated steam at bar absolute enters the heating coil and saturated liquid at the same pressure exits the heating coil, with the latent heat evaporating water in the process.
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


