Answered step by step
Verified Expert Solution
Question
1 Approved Answer
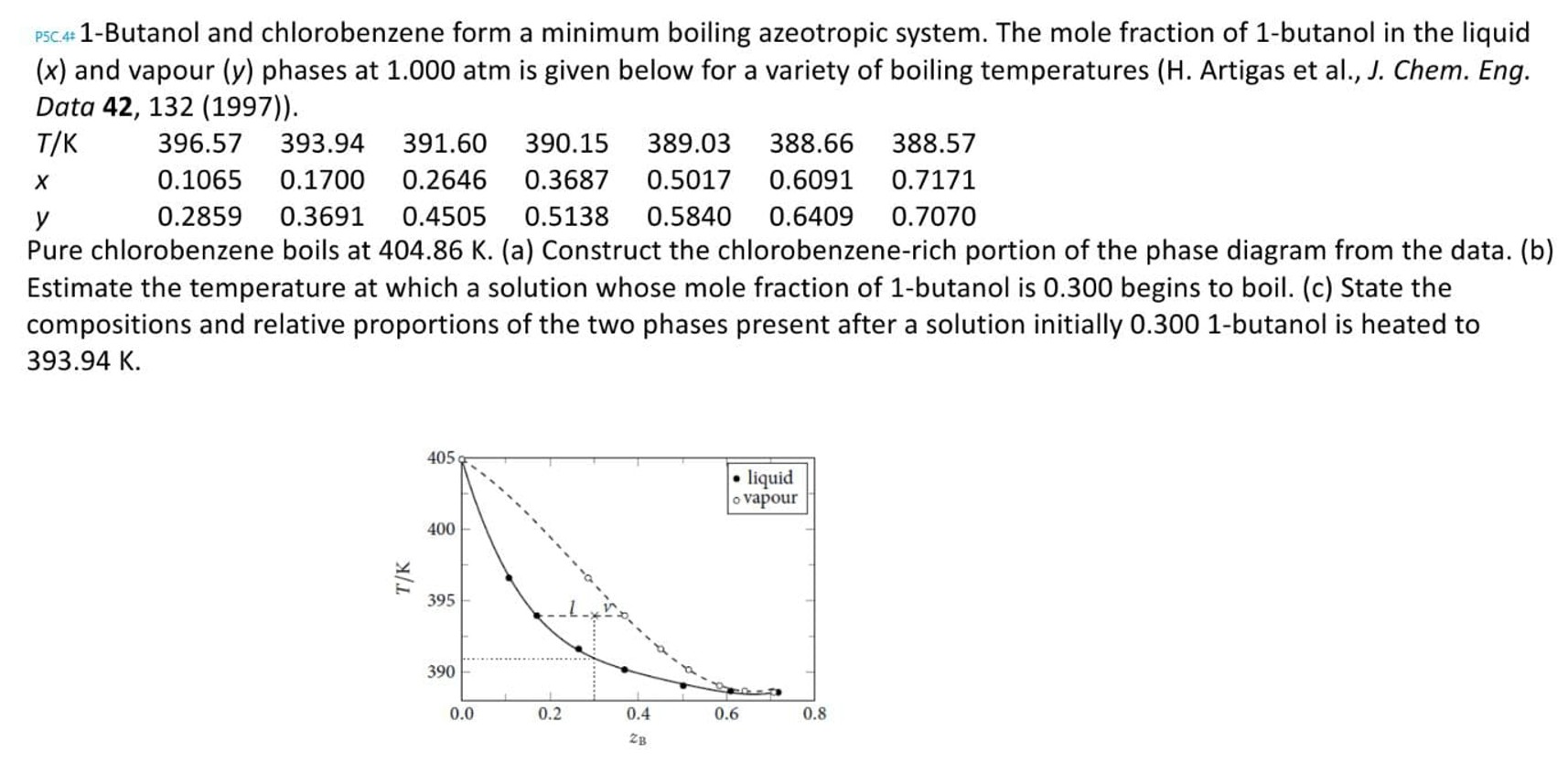
? P 5 C . 4 1 - Butanol and chlorobenzene form a minimum boiling azeotropic system. The mole fraction of 1 - butanol in
Butanol and chlorobenzene form a minimum boiling azeotropic system. The mole fraction of butanol in the liquid
and vapour phases at atm is given below for a variety of boiling temperatures H Artigas et al J Chem. Eng.
Pure chlorobenzene boils at a Construct the chlorobenzenerich portion of the phase diagram from the data. b
Estimate the temperature at which a solution whose mole fraction of butanol is begins to boil. c State the
compositions and relative proportions of the two phases present after a solution initially butanol is heated to
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


