Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Part B: Equilibrium Test Solutions for the Determination of Kna Table 3: Sot of Equilibrium Test Solutions for the Determination of Kon Use these data
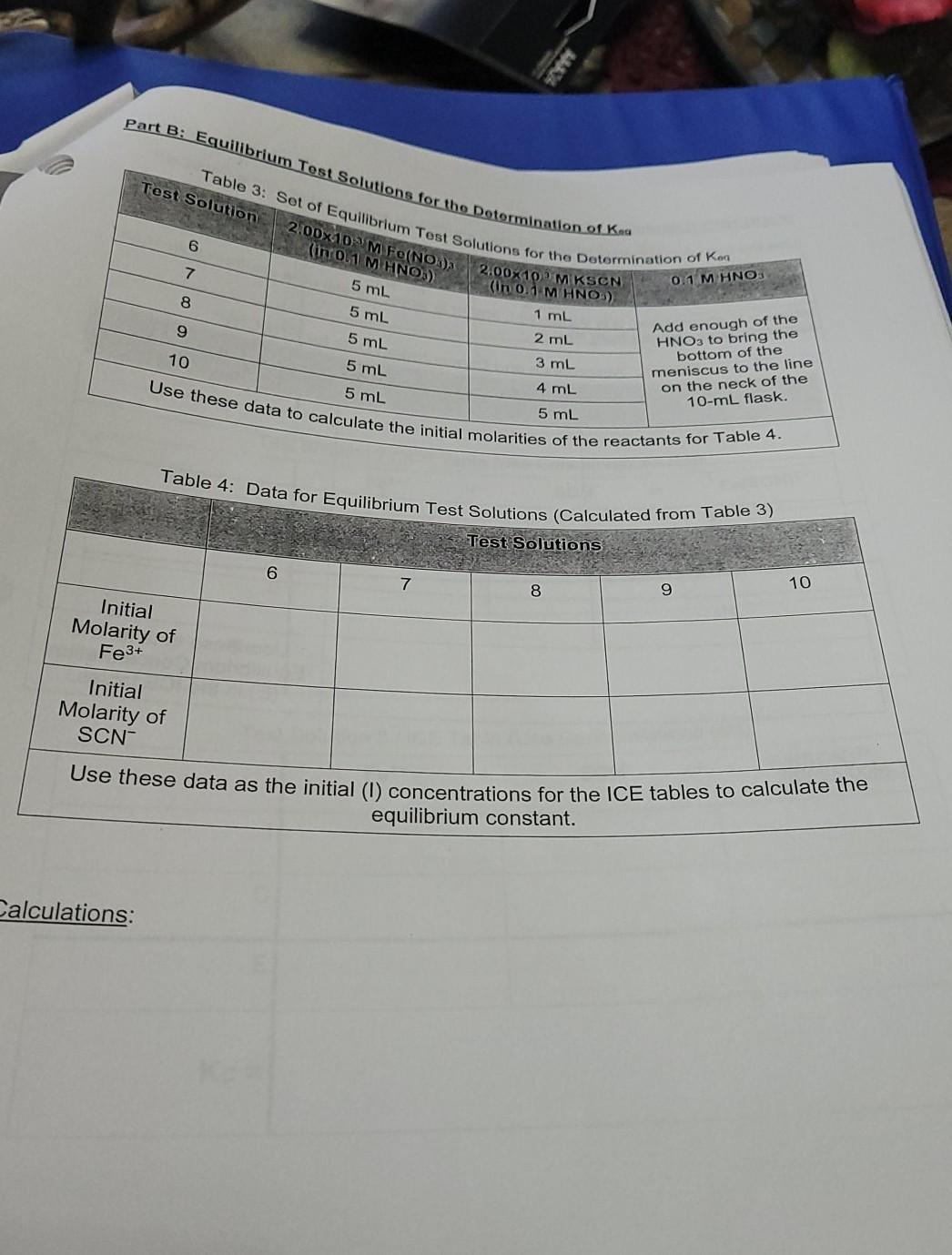
Part B: Equilibrium Test Solutions for the Determination of Kna Table 3: Sot of Equilibrium Test Solutions for the Determination of Kon Use these data to calculate the initial molarities of the reactants for Table 4. Table 4: Data for Equilibrium Test Solutions (Calculated from Table 3) Test Solution 6 2.00x10 M Fe(NO); (in o.1 M HNO) 5 ml 7 2.00x 10 MKSCN (in 0.1 M HNO) 0.1 MHNO. 8 5 mL 1 ml 9 5 ML 2 mL 3 mL 10 5 mL Add enough of the HNO3 to bring the bottom of the meniscus to the line on the neck of the 10-ml flask. 5 mL 4 mL 5 mL Test Solutions 6 7 8 9 10 Initial Molarity of Fe3+ Initial Molarity of SCN- Use these data as the initial (1) concentrations for the ICE tables to calculate the equilibrium constant. Calculations
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


