Answered step by step
Verified Expert Solution
Question
1 Approved Answer
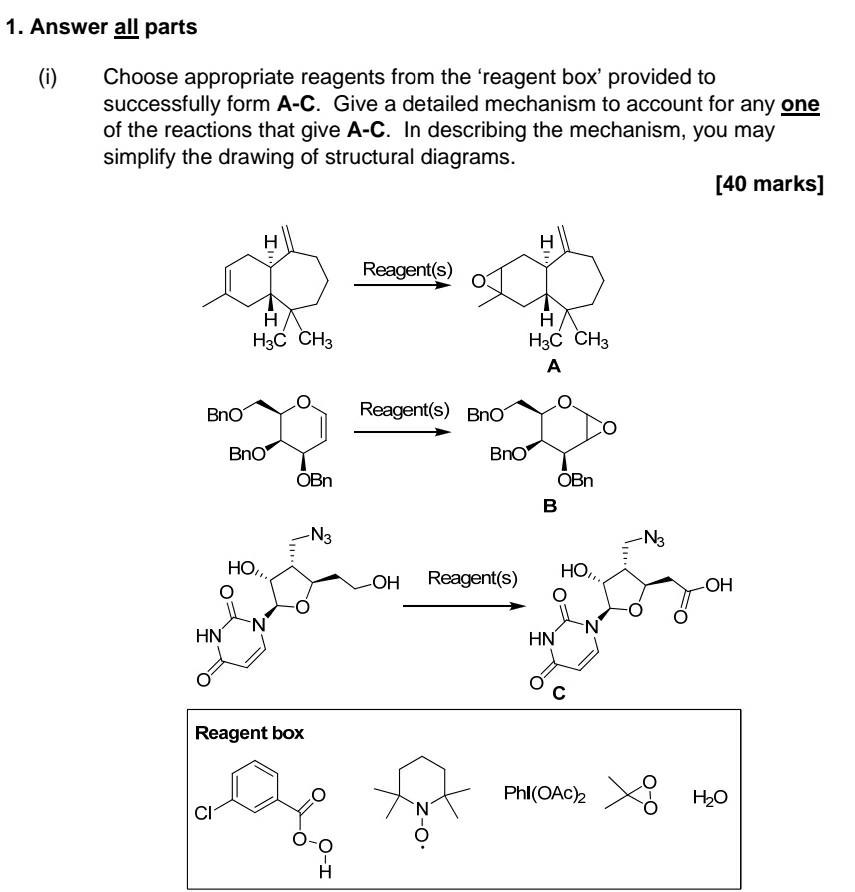
Part i) Part ii) & iii) 1. Answer all parts (i) Choose appropriate reagents from the 'reagent box' provided to successfully form A-C. Give a
Part i)
Part ii) & iii)
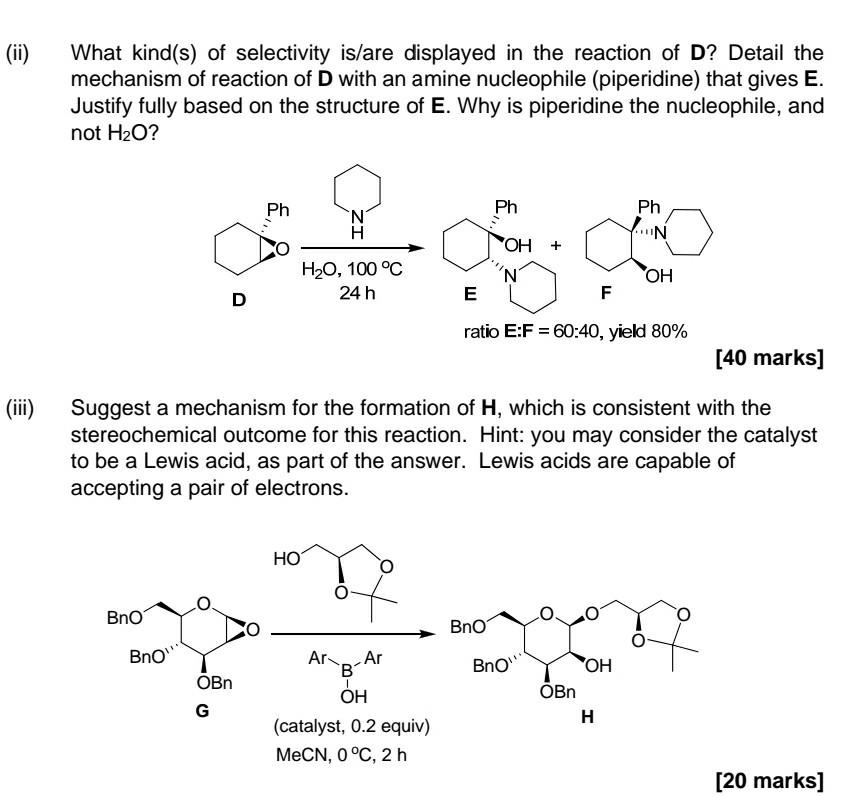
1. Answer all parts (i) Choose appropriate reagents from the 'reagent box' provided to successfully form A-C. Give a detailed mechanism to account for any one of the reactions that give A-C. In describing the mechanism, you may simplify the drawing of structural diagrams. [40 marks] Reagent(s) H3 C CH3 H H3C CH3 A Bno Reagent(s) Bno Bno OBn Bno OBn B - -N -N3 HO, , , -OH Reagent(s) - N N HN HN Reagent box Phl(OAC)2 H2O CI N . (ii) What kind(s) of selectivity is/are displayed in the reaction of D? Detail the mechanism of reaction of D with an amine nucleophile (piperidine) that gives E. Justify fully based on the structure of E. Why is piperidine the nucleophile, and not H2O? Ph Ph Ph N H IN o OH + H20, 100 C 24 h 'N OH D E F ratio E:F = 60:40, yield 80% [40 marks] (iii) Suggest a mechanism for the formation of H, which is consistent with the stereochemical outcome for this reaction. Hint: you may consider the catalyst to be a Lewis acid, as part of the answer. Lewis acids are capable of accepting a pair of electrons. HO BnO Bn01 Bn0 Bn0 1 OBn G Ar Ar B OH (catalyst, 0.2 equiv) MeCN, 0 C, 2 h OH OBn H [20 marks]
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


