Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Pease answer a&b with work shown. The phosphate buffer solution has a ph=7.5, pka=2.13, and (A-/HA) ratio= 2.4x10^5 1. This problem will be done for
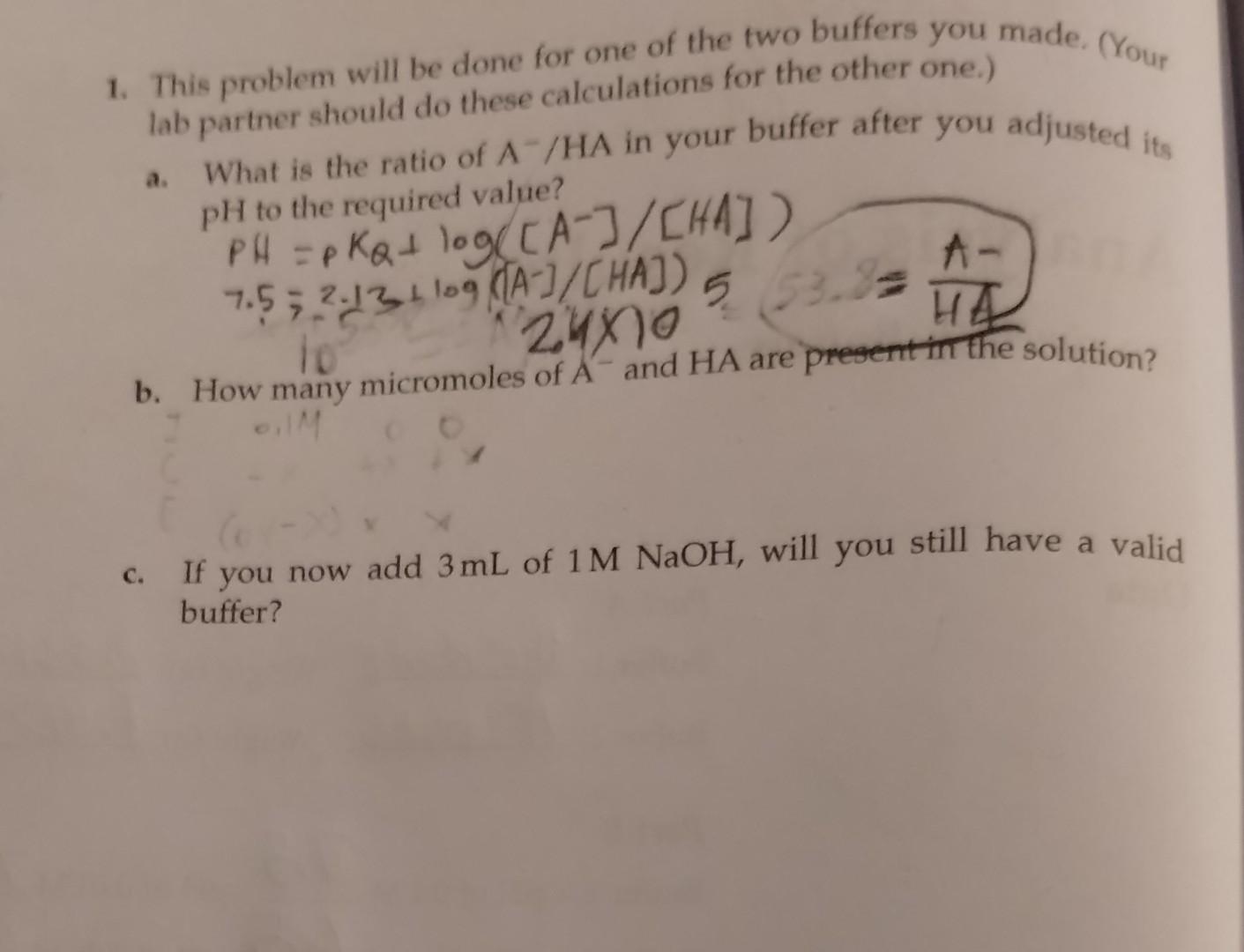
Pease answer a&b with work shown. The phosphate buffer solution has a ph=7.5, pka=2.13, and (A-/HA) ratio= 2.4x10^5
1. This problem will be done for one of the two buffers you made. (Your lab partner should do these calculations for the other one.) a. What is the ratio of A/HA in your buffer after you adjusted its PH to the required value? Pf=PKQ+log([A]/[HA]) 7.52.13+log([A]/[HA])5 b. How many micromoles of Aand HA are present in the solution? c. If you now add 3mL of 1MNaOH, will you still have a valid buffer? 1. This problem will be done for one of the two buffers you made. (Your lab partner should do these calculations for the other one.) a. What is the ratio of A/HA in your buffer after you adjusted its PH to the required value? Pf=PKQ+log([A]/[HA]) 7.52.13+log([A]/[HA])5 b. How many micromoles of Aand HA are present in the solution? c. If you now add 3mL of 1MNaOH, will you still have a valid bufferStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


