Answered step by step
Verified Expert Solution
Question
1 Approved Answer
please answee 7-12 7. Which substance would you expect to experience dipole-dipole intermolecular forces? A) thy B) CCl4 c) NF3 D) CS2 E) none of
please answee 7-12 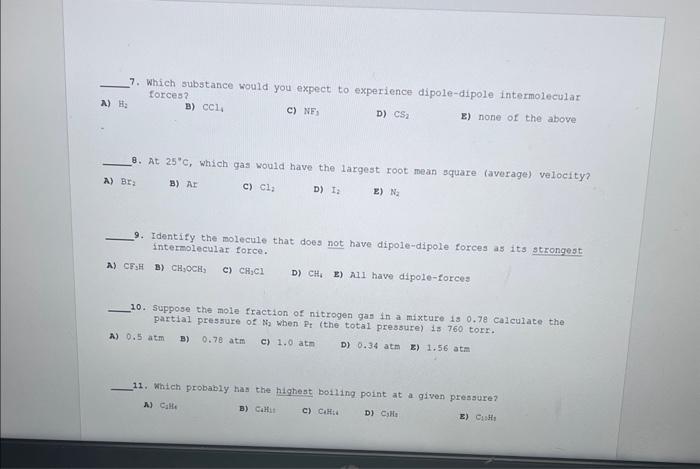

7. Which substance would you expect to experience dipole-dipole intermolecular forces? A) thy B) CCl4 c) NF3 D) CS2 E) none of the above 8. At 25C, which gas would have the largest root mean square (average) velocity? A) Br2 B) NI C) Cl2 D) I2 E) Na2 9. Identify the molecule that does not have dipole-dipole forces as its strongest interelecular force. A) CF3H B) CH2CCH3 c) CH1Cl D) CH6 E) Ali have dipole-forces 10. Suppose the mole fraction of nitrogen gas in a mixture 150.78 calculate the partial pressure of N2 when Pt (the total pressure) 1$760 torr. A) 0.5atm B) 0.78atm c) 1.0atm D) 0.34atm z) 1.56atm 11. Which probably has the highest boiling point at a given pressure? ) Calfe B) CaHit c) Culs:i D) CiH4 a) C1:H1 12. What is the order of increasing density of the following gases at the same temperature and pressure? N2,NH3,N2O6, and Ar A) N2O4

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


