Answered step by step
Verified Expert Solution
Question
1 Approved Answer
please answer all parts to number 1. attached is the table for standard reduction potentials 1. Consider the following electrochemical cell at 25C : Fe(s)FeCl3(aq)Cl2(g)Pt
please answer all parts to number 1. attached is the table for standard reduction potentials 
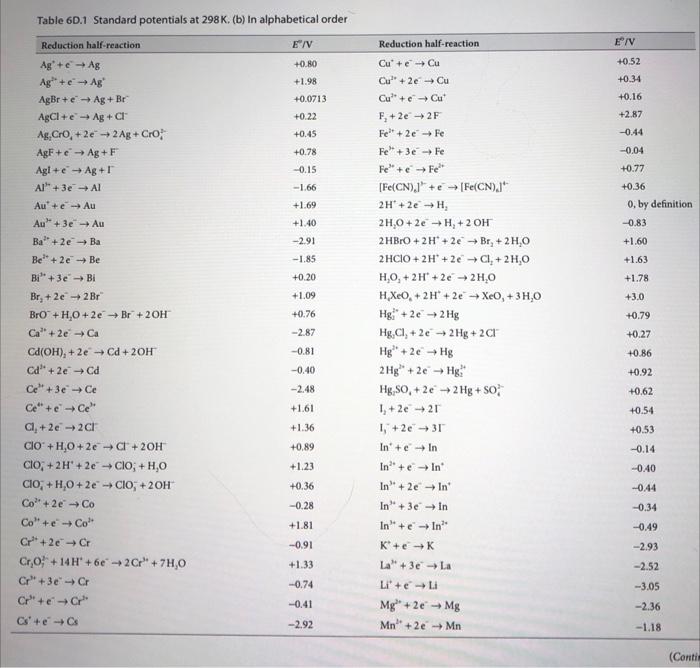
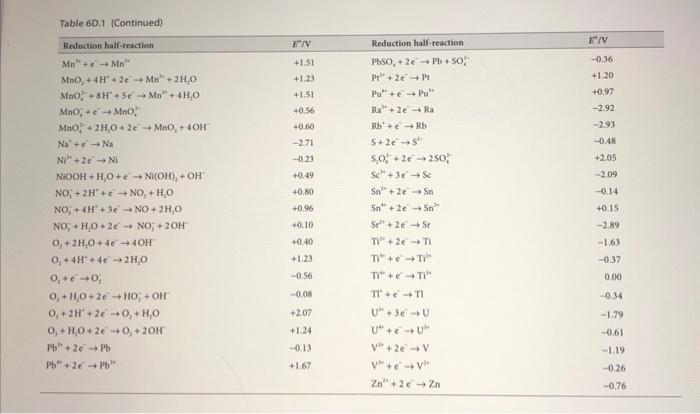
1. Consider the following electrochemical cell at 25C : Fe(s)FeCl3(aq)Cl2(g)Pt a) Write each half-reaction then write the complete electrochemical reaction for this cell. Neglect any complex ion formation between Fe3+ and Clions. b) Using the table of standard reduction potentials in the text, calculate E for this cell. c) What is the equilibrium constant for the chemical reaction in part (a)? d) What is G for the chemical reaction of this cell? e) If the Cl2 pressure is 1.5bar, and the FeCl3 concentration is 0.020m, what is E for the cell? Use the Davies equation to estimate for FeCl3. Assume the Cl2 behaves as an ideal gas. f) Which direction will electrons spontaneously flow if the Fe terminal is connected to the Pt at the conditions of part(e)? Table 6D.1 Standard potentials at 298K. (b) In alphabetical order E/V+0.52+0.34+0.16+2.870.440.04+0.77+0.360,bydefinition0.83+1.60+1.63+1.78+3.0+0.79+0.27+0.86+0.92+0.62+0.54+0.530.140.400.440.340.492.932.523.052.361.18 Table 6D.1 (Continued) 


Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


