Answered step by step
Verified Expert Solution
Question
1 Approved Answer
please answer thanks Under certain conditions the rate of this reaction is zero order in ammonia with a rate constant of 0.0016.M1 : : 2NH3(g)N2(g)+3H2(g)
please answer 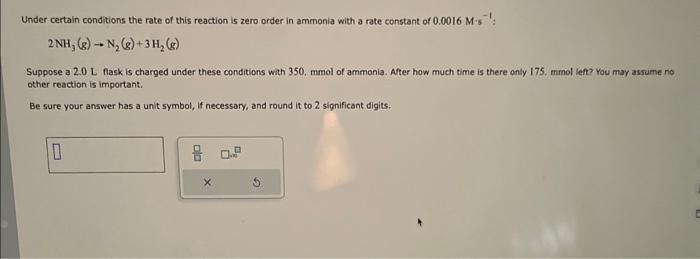
Under certain conditions the rate of this reaction is zero order in ammonia with a rate constant of 0.0016.M1 : : 2NH3(g)N2(g)+3H2(g) Suppose a 2.0L flask is charged under these conditions with 350 , mmol of ammonia. After how much time is there only 175 , thmol left? You may assume no other reaction is important. Be sure your answer has a unit symbol, if necessary, and round it to 2 significant digits thanks

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


