Answered step by step
Verified Expert Solution
Question
1 Approved Answer
please answer what you can! volume at equivalence point Volume at one-half equivalence point pKa Ka Average Ka Standard Deviation DATA ANALYSIS ml NaOH PH
please answer what you can!
DATA ANALYSIS ml NaOH PH First Determination 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 3.24 3.28 3.48 3.63 3.75 3.86 3.96 4.00 4.04 4.10 4.13 4.20 427 4.33 4.37 4.41 4.46 4.52 4.56 4.62 4.67 4.23 4.80 4.85 4.91 4.99 512 5120 5:30 5.51 59 10.24 10.87 pH Second Determination 3.19 3.33 3.51 3.64 3.77 3.86 3.94 4.01 4.08 4.13 4.19 4.25 4.29 4.35 4.37 443 4.47 4.51 4.55 4.60 4.65 4.71 4.76 4.82 4.88 4.97 5.04 pH Third Determination 3.38 3.43 3.58 3.69 3.80 3.89 3.95 4,02 4.07 4.13 4.19 4.24 4.29 4.33 4.36 4.41 4.45 4.49 4.53 4.58 4.62 4.67 4.72 4.76 4.82 4.92 4.97 5110 5.20 5.44 5.70 10.2 11.11 5.14 5.27 5.40 5.61 10155 11.04 Volume at equivalence point: Volume at one-half equivalence point Ka Average Ka= Standard Deviation = volume at equivalence point 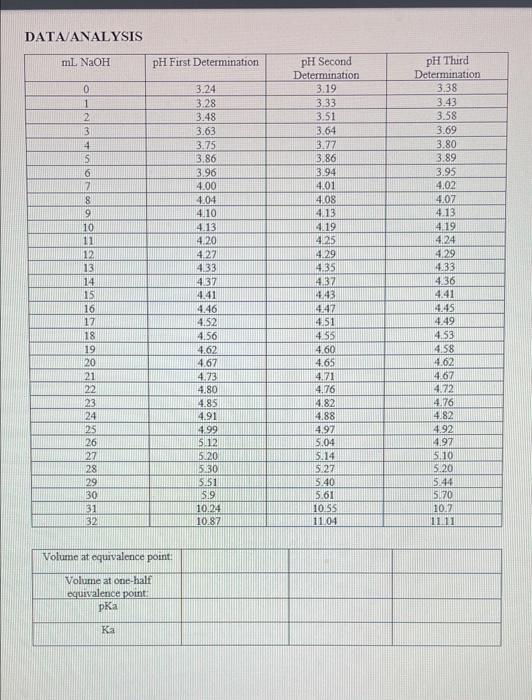
Volume at one-half equivalence point
pKa
Ka
Average Ka
Standard Deviation

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


