Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Please explain thank you! PFe(0) PFe+0, 3. Consider the interchange of O2 and CO coordinated to myoglobin, an iron porphyrin denoted as PFe: ; PFe
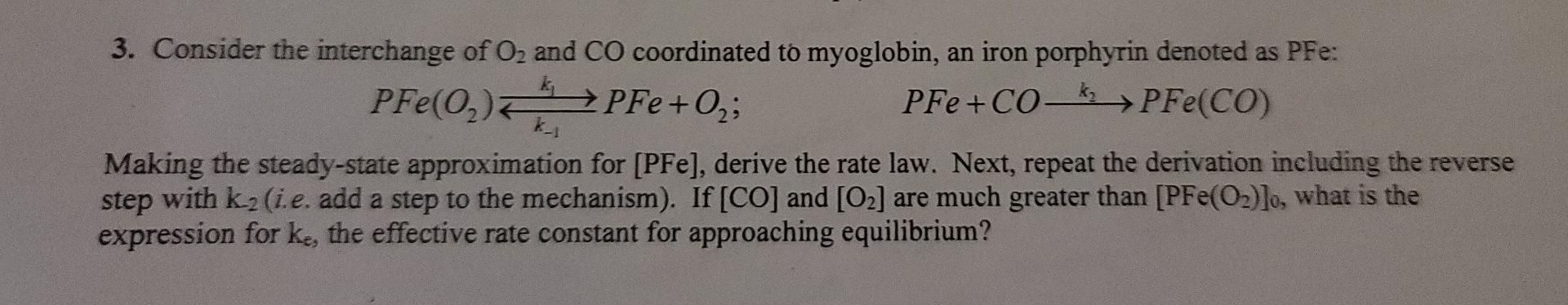
Please explain thank you!
PFe(0) PFe+0, 3. Consider the interchange of O2 and CO coordinated to myoglobin, an iron porphyrin denoted as PFe: ; PFe +CO PFe(CO) k_1 Making the steady-state approximation for [PFe), derive the rate law. Next, repeat the derivation including the reverse step with k.(i.e. add a step to the mechanism). If [CO] and [O2] are much greater than [PFe(O2)]o, what is the expression for ke, the effective rate constant for approaching equilibriumStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


