Answered step by step
Verified Expert Solution
Question
1 Approved Answer
please explain with a graph. Below the data is shown for reversible cis-trans isomerization of 1,2-dimethyl cyclopropane. Test the data to ascertain if they are
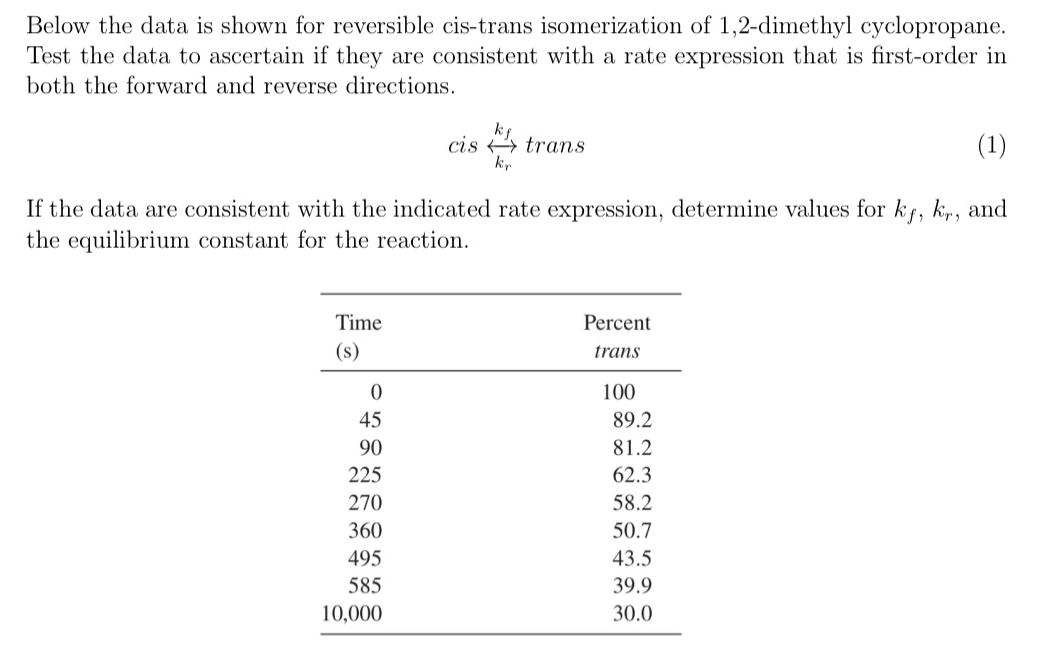
please explain with a graph.
Below the data is shown for reversible cis-trans isomerization of 1,2-dimethyl cyclopropane. Test the data to ascertain if they are consistent with a rate expression that is first-order in both the forward and reverse directions. ciskrkftrans If the data are consistent with the indicated rate expression, determine values for kf,kr, and the equilibrium constant for the reaction. Below the data is shown for reversible cis-trans isomerization of 1,2-dimethyl cyclopropane. Test the data to ascertain if they are consistent with a rate expression that is first-order in both the forward and reverse directions. ciskrkftrans If the data are consistent with the indicated rate expression, determine values for kf,kr, and the equilibrium constant for the reactionStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


