Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Please explain your steps and I will give your answer a like! Problem 3 The equation below gives the density in kg/L of formic acid
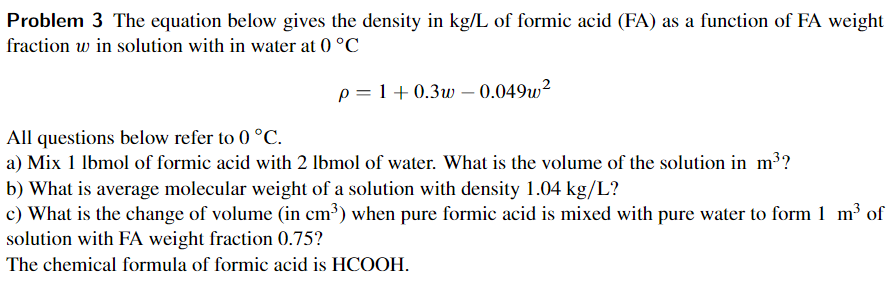
Please explain your steps and I will give your answer a like!
Problem 3 The equation below gives the density in kg/L of formic acid (FA) as a function of FA weight fraction w in solution with in water at 0 C p=1+0.3w - 0.049w2 All questions below refer to () C. a) Mix 1 lbmol of formic acid with 2 lbmol of water. What is the volume of the solution in m?? b) What is average molecular weight of a solution with density 1.04 kg/L? c) What is the change of volume (in cm?) when pure formic acid is mixed with pure water to form 1 m of solution with FA weight fraction 0.75? The chemical formula of formic acid is HCOOHStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


