Answered step by step
Verified Expert Solution
Question
1 Approved Answer
PLEASE HELP ASAP WILL LIKE! 1. Identify the hybridization state and geometry of each non-hydrogen atom in the following structure of lysergic acid ethyldiamide (LSD),
PLEASE HELP ASAP WILL LIKE! 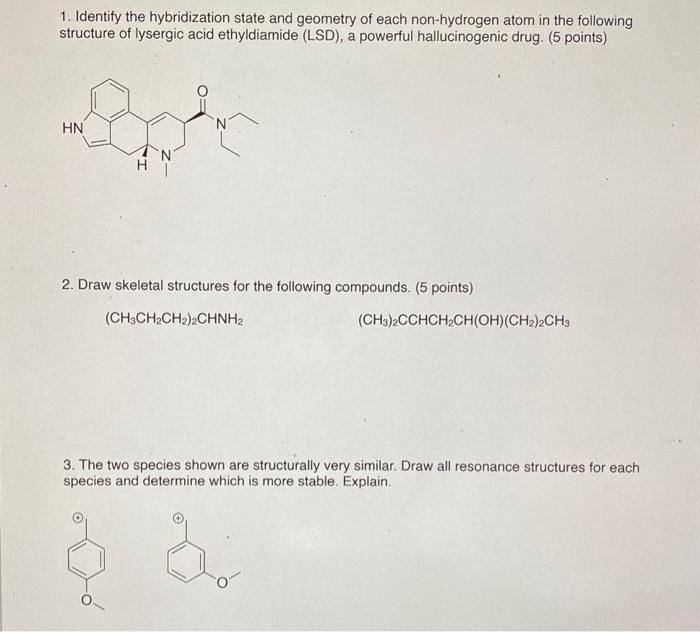
1. Identify the hybridization state and geometry of each non-hydrogen atom in the following structure of lysergic acid ethyldiamide (LSD), a powerful hallucinogenic drug. (5 points) 2. Draw skeletal structures for the following compounds. ( 5 points) (CH3CH2CH2)2CHNH2 (CH3)2CCHCH2CH(OH)(CH2)2CH3 3. The two species shown are structurally very similar. Draw all resonance structures for each species and determine which is more stable. Explain 
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


