Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Please help. thank you! Calculate Kp for this reaction at 500.0K. Express your answer using two significant figures. The equilibrium 2NO(g)+Cl2(g)2NOCl(g) is established at 500K.
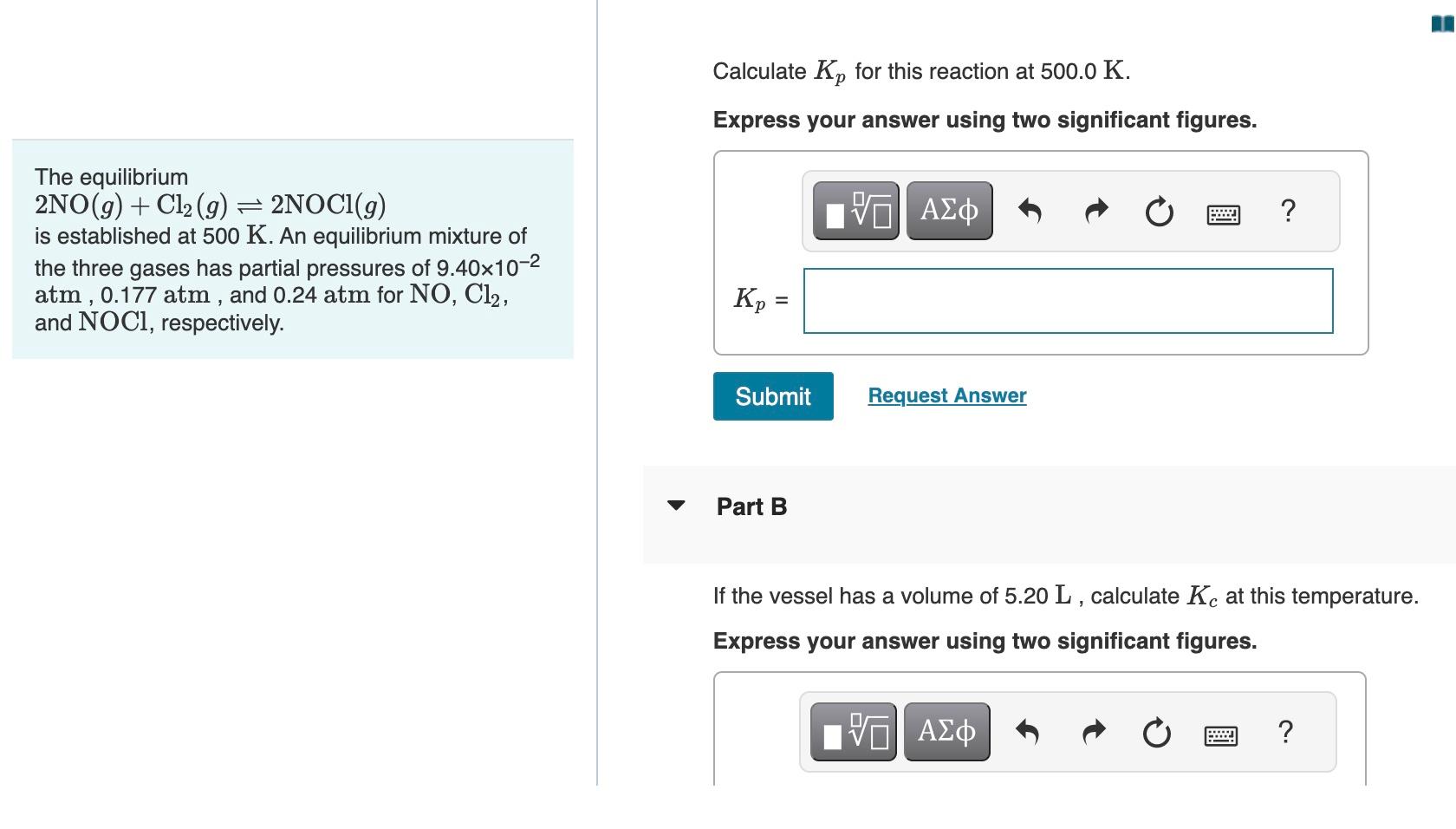
Please help. thank you!
Calculate Kp for this reaction at 500.0K. Express your answer using two significant figures. The equilibrium 2NO(g)+Cl2(g)2NOCl(g) is established at 500K. An equilibrium mixture of the three gases has partial pressures of 9.40102 atm , 0.177atm, and 0.24atm for NO,Cl2, and NOCl, respectively. Part B If the vessel has a volume of 5.20L, calculate Kc at this temperature. Express your answer using two significant figuresStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


