Answered step by step
Verified Expert Solution
Question
1 Approved Answer
please help with all 3! 1. On a nice day, 28.3C, the volume of your gummy bear pool float is 15,072in3. As Fall approaches in
please help with all 3! 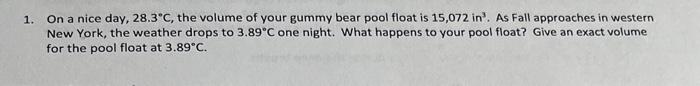



1. On a nice day, 28.3C, the volume of your gummy bear pool float is 15,072in3. As Fall approaches in western New York, the weather drops to 3.89C one night. What happens to your pool float? Give an exact volume for the pool float at 3.89C. Calculate the volume (in mL ) that a helium gas sample would occupy under STP, if that same helium gas sample occupies 437mL under a pressure of 783mmHg at 179C. 3. In order to inhale the diaphragm contracts (flattens out) to increase the volume of the thoracic cavity, which will allow the lungs to expand. The shift by the diaphragm allows a large rush of air to be inhaled due to the lowered pressure in the thoracic cavity compared to the outside pressure. At any given moment the lung capacity is 2570.mL of air at 760.mmHg, but when the diaphragm contracts the volume inside of the cavity shifts to 3070.mL. a. Calculate the pressure in thoracic cavity during this deep breath. b. Does it surprise you to know the actual change in pressure by the thoracic cavity is 4mmHg ? If yes, give a suggestion as to why the difference 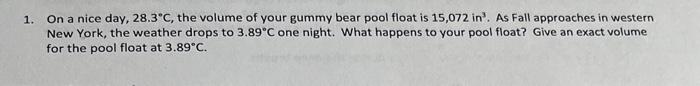



Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


