Answered step by step
Verified Expert Solution
Question
1 Approved Answer
please help with all three Acetylene (C2H2), an important fuel in welding, is produced in the laboratory when calcium carbide (CaC2) reacts with water: CaC2(s)+2H2O(l)C2H2(g)+Ca(OH)2(aq)
please help with all three 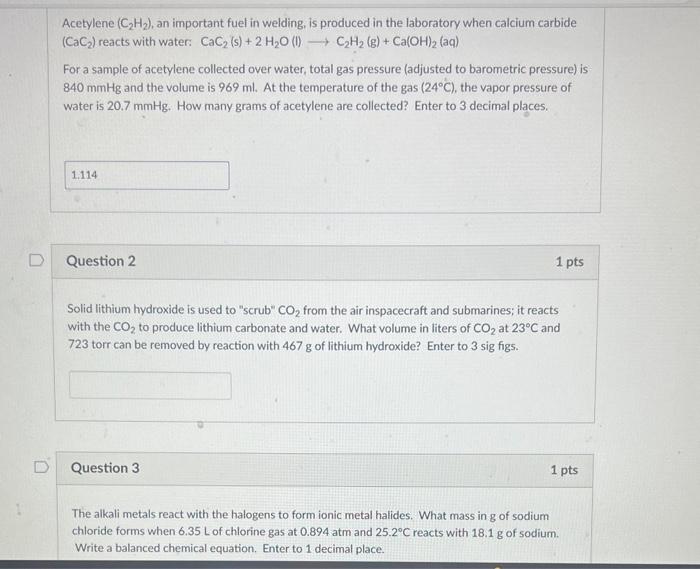
Acetylene (C2H2), an important fuel in welding, is produced in the laboratory when calcium carbide (CaC2) reacts with water: CaC2(s)+2H2O(l)C2H2(g)+Ca(OH)2(aq) For a sample of acetylene collected over water, total gas pressure (adjusted to barometric pressure) is 840mmHg and the volume is 969ml. At the temperature of the gas (24C), the vapor pressure of water is 20.7mmHg. How many grams of acetylene are collected? Enter to 3 decimal places. Question 2 1 pts Solid lithium hydroxide is used to "scrub" CO2 from the air inspacecraft and submarines; it reacts with the CO2 to produce lithium carbonate and water. What volume in liters of CO2 at 23C and 723 torr can be removed by reaction with 467g of lithium hydroxide? Enter to 3 sig figs. Question 3 1 pts The alkali metals react with the halogens to form ionic metal halides. What mass in g of sodium chloride forms when 6.35L of chlorine gas at 0.894atm and 25.2C reacts with 18.1g of sodium. Write a balanced chemical equation, Enter to 1 decimal place 
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


