Answered step by step
Verified Expert Solution
Question
1 Approved Answer
please help with this thanks Calculate Kea. (2) Consider: SiH4(g)+2Cl2(g)SiCl4(g)+2H2(g) The gases Cl2 and SiH4 are placed into a 1.0L reaction flask and allowed to
please help with this thanks 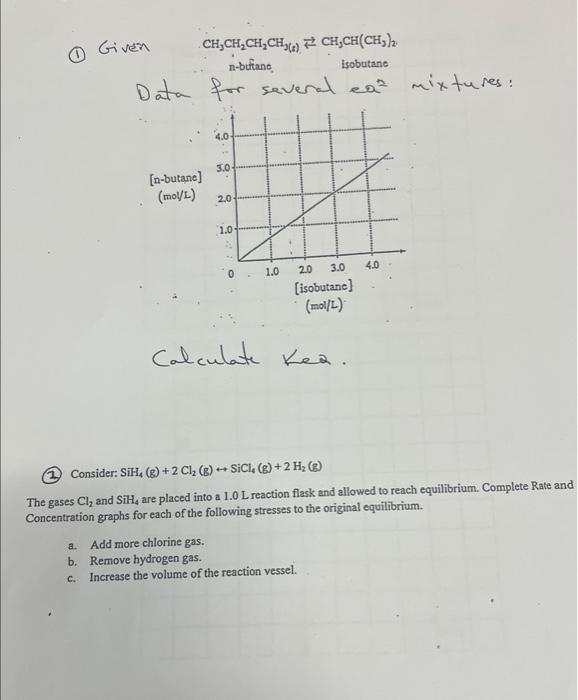
Calculate Kea. (2) Consider: SiH4(g)+2Cl2(g)SiCl4(g)+2H2(g) The gases Cl2 and SiH4 are placed into a 1.0L reaction flask and allowed to reach equilibrium. Complete Rate and Concentration graphs for each of the following stresses to the original equilibrium. a. Add more chlorine gas. b. Remove hydrogen gas. c. Increase the volume of the reaction vessel 
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


