Answered step by step
Verified Expert Solution
Question
1 Approved Answer
pls help Calculate solution concentration A solution consists of 44.3 g of ammonia, NH3, and 103.7 g water. (a) Calculate the weight percent, the molality,
pls help 
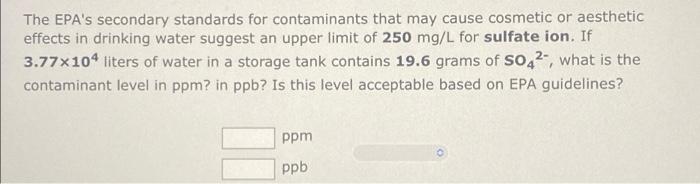
Calculate solution concentration A solution consists of 44.3 g of ammonia, NH3, and 103.7 g water. (a) Calculate the weight percent, the molality, and the mole fraction of NH3 in the solution. % weight percent = molality = mole fraction = m (b) The solution volume is 166 ml. Calculate the molarity of NH3 in the solution. molarity M The EPA's secondary standards for contaminants that may cause cosmetic or aesthetic effects in drinking water suggest an upper limit of 250 mg/L for sulfate ion. If 3.77x104 liters of water in a storage tank contains 19.6 grams of s042, what is the contaminant level in ppm? in ppb? Is this level acceptable based on EPA guidelines? ppm ppb 

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


