Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Pls show detailed working on which cell to use (Eg.: B17*B18*) to get the Length of packed bed required for the reactor and the final
Pls show detailed working on which cell to use (Eg.: B17*B18*) to get the Length of packed bed required for the reactor and the final answer using the 4th order Runge Kutta method.
Note: Pls provide the Excel sheet and a screenshot of it as well.
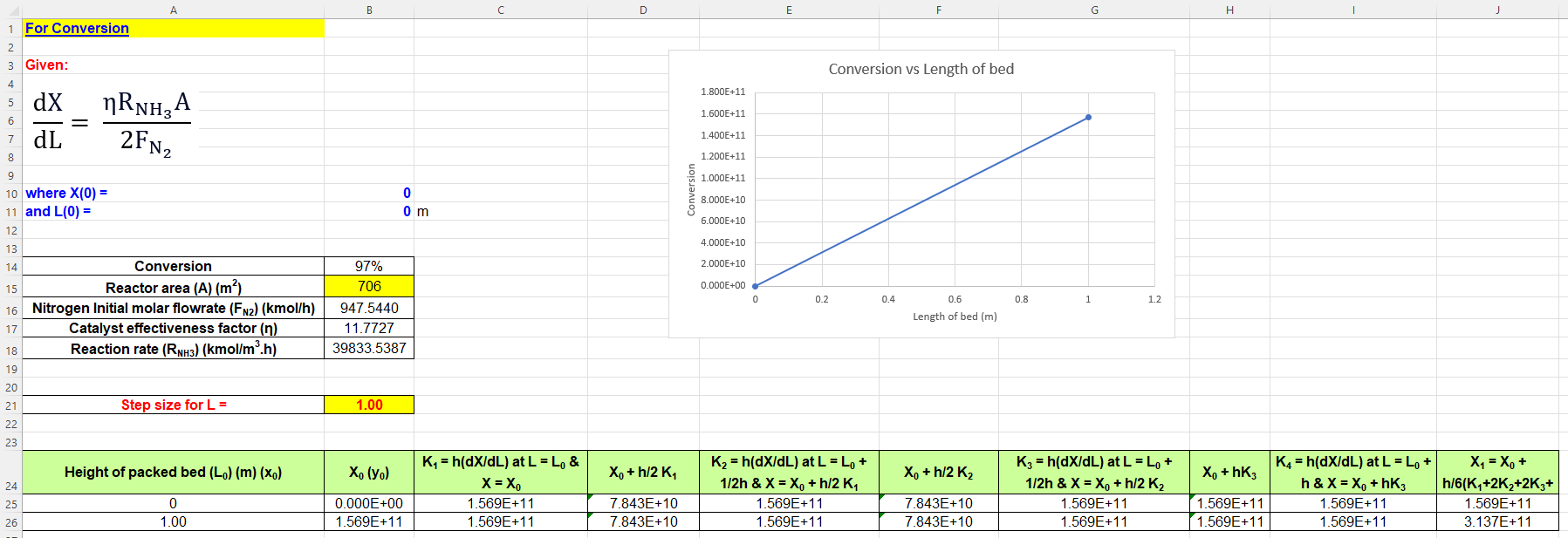
For Conversion Given: dLdX=2FN2RNH3A where X(0)= and L(0)= 0 0 \begin{tabular}{|c|c|} \hline Conversion & 97% \\ \hline Reactor area (A)(m2) & 706 \\ \hline Nitrogen Initial molar flowrate (FN2)(kmol/h) & 947.5440 \\ \hline Catalyst effectiveness factor () & 11.7727 \\ \hline Reaction rate (RNH3)(kmol/m3.h) & 39833.5387 \\ \hline \end{tabular} Conversion vs Length of bed 1.800E+11 Step size for L= 1.00 Length of bed (m) H For Conversion Given: dLdX=2FN2RNH3A where X(0)= and L(0)= 0 0 \begin{tabular}{|c|c|} \hline Conversion & 97% \\ \hline Reactor area (A)(m2) & 706 \\ \hline Nitrogen Initial molar flowrate (FN2)(kmol/h) & 947.5440 \\ \hline Catalyst effectiveness factor () & 11.7727 \\ \hline Reaction rate (RNH3)(kmol/m3.h) & 39833.5387 \\ \hline \end{tabular} Conversion vs Length of bed 1.800E+11 Step size for L= 1.00 Length of bed (m) HStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


