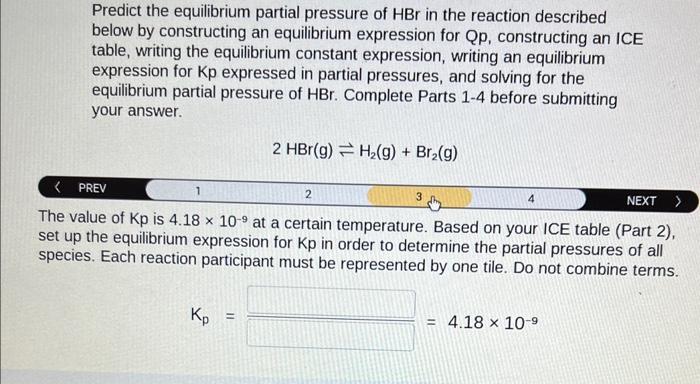
Predict the equilibrium partial pressure of HBr in the reaction described below by constructing an equilibrium expression for Qp, constructing an ICE table, writing the equilibrium constant expression, writing an equilibrium expression for Kp expressed in partial pressures, and solving for the equilibrium partial pressure of HBr. Complete Parts 1-4 before submitting your answer. 2HBr(g)H2(g)+Br2(g) An initial mixture of 0.30atmHBr,0.010atmH2, and 0.010atmBr2 reacts. Set up the expression for Qp to determine the direction of the reaction. Each reaction participant must be represented by one tile. Do not combine terms. Once the expression is constructed, solve for Qp. Qp= Predict the equilibrium partial pressure of HBr in the reaction described below by constructing an equilibrium expression for Qp, constructing an ICE table, writing the equilibrium constant expression, writing an equilibrium expression for Kp expressed in partial pressures, and solving for the equilibrium partial pressure of HBr. Complete Parts 1-4 before submitting your answer. 2HBr(g)H2(g)+Br2(g) PREV 1 3 NEXT > Based on the initial pressure, your value of Qp, and that Kp is 4.18109, fill in the ICE table with the appropriate value for each involved species to determine the partial pressures of all reactants and products. Predict the equilibrium partial pressure of HBr in the reaction described below by constructing an equilibrium expression for Qp, constructing an ICE table, writing the equilibrium constant expression, writing an equilibrium expression for Kp expressed in partial pressures, and solving for the equilibrium partial pressure of HBr. Complete Parts 1-4 before submitting your answer. 2HBr(g)H2(g)+Br2(g) \begin{tabular}{l|cccccc} PREV & 1 & 2 & 4 & NEXT \end{tabular} set up the equilibrium expression for Kp in order to determine the partial pressures of all species. Each reaction participant must be represented by one tile. Do not combine terms. Kp= Predict the equilibrium partial pressure of HBr in the reaction described below by constructing an equilibrium expression for Qp, constructing an ICE table, writing the equilibrium constant expression, writing an equilibrium expression for Kp expressed in partial pressures, and solving for the equilibrium partial pressure of HBr. Complete Parts 1-4 before submitting your answer. 2HBr(g)H2(g)+Br2(g) HBr at equilibrium. PHBr=atm