Answered step by step
Verified Expert Solution
Question
1 Approved Answer
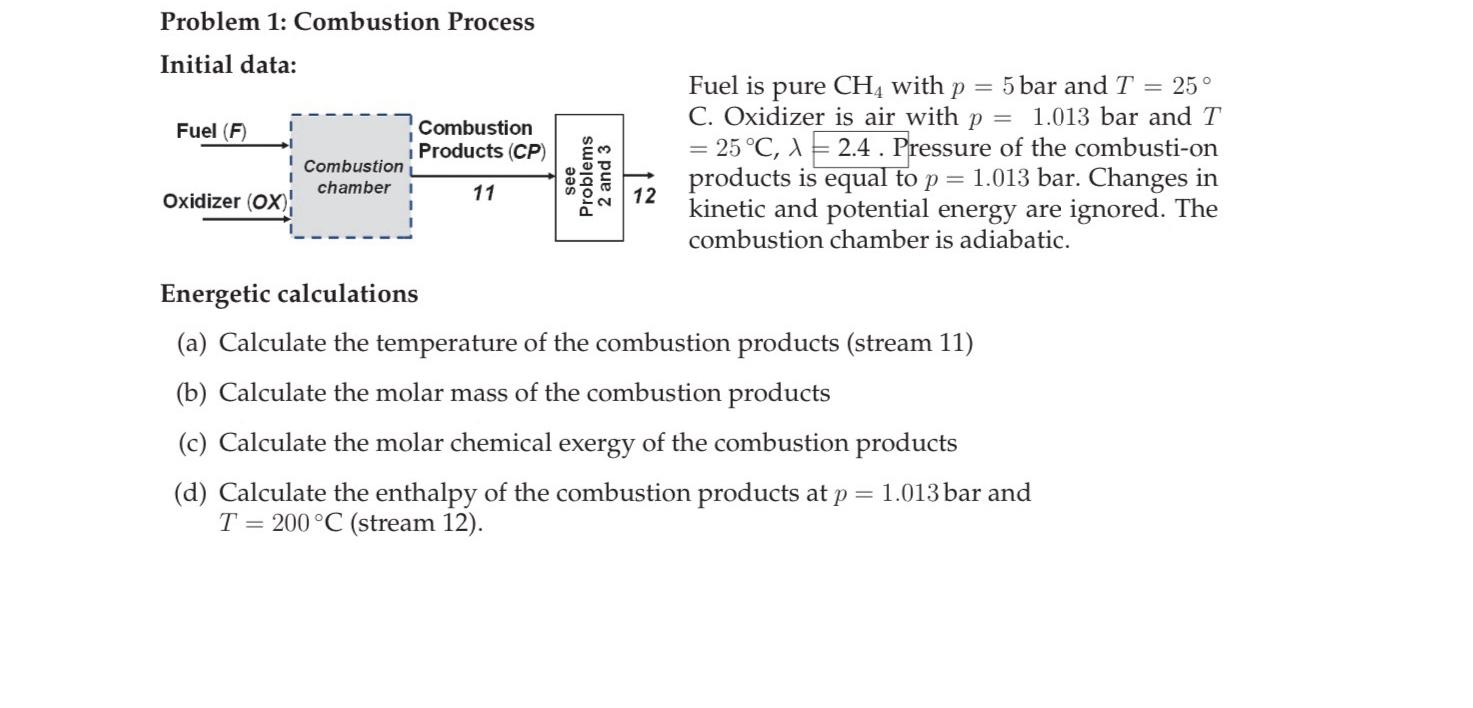
Problem 1 : Combustion Process Initial data: Fuel is pure C H 4 with p = 5 bar and T = 2 5 C .
Problem : Combustion Process
Initial data:
Fuel is pure with bar and
C Oxidizer is air with bar and
Pressure of the combustion products is equal to bar. Changes in kinetic and potential energy are ignored. The combustion chamber is adiabatic.
Energetic calculations
a Calculate the temperature of the combustion products stream
b Calculate the molar mass of the combustion products
c Calculate the molar chemical exergy of the combustion products
d Calculate the enthalpy of the combustion products at bar and stream
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


