Question
1. a. Which of the following components has the highest vapor pressure? b. Which of the components below dissolves best in hexane? Justify your
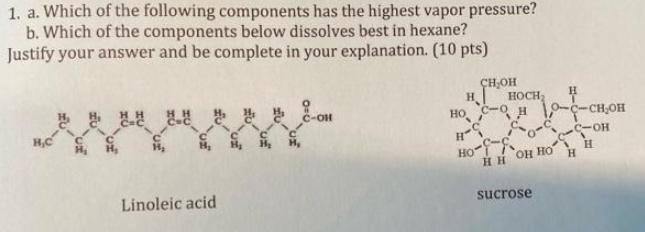
1. a. Which of the following components has the highest vapor pressure? b. Which of the components below dissolves best in hexane? Justify your answer and be complete in your explanation. (10 pts) H,C Linoleic acid C-OH CHOH H HOCH H - 10-C-CHOH H-S -- Ho H HH OH HO sucrose H
Step by Step Solution
3.39 Rating (155 Votes )
There are 3 Steps involved in it
Step: 1
Answer Linoleic acid has the highest vapor pressure of the two components This is because it is the lightest and has the lowest boiling point Sucrose ...
Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get StartedRecommended Textbook for
The Physics of Energy
Authors: Robert L. Jaffe, Washington Taylor
1st edition
1107016657, 978-1107016651
Students also viewed these Physics questions
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
View Answer in SolutionInn App



