Question: Q8: Below is a unit cell for a hypothetical metal. (a) To which crystal system does this unit cell belong? (b) What would this crystal
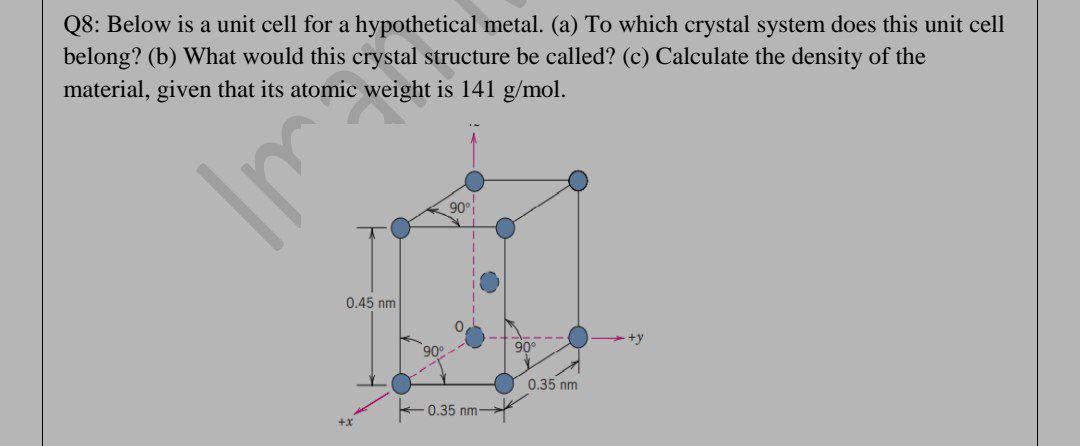
Q8: Below is a unit cell for a hypothetical metal. (a) To which crystal system does this unit cell belong? (b) What would this crystal structure be called? (c) Calculate the density of the material, given that its atomic weight is 141 g/mol. 90 C 0.45 nm 90 90 0.35 nm 0.35 nm- +
Step by Step Solution
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


