Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Question 1 part (a) part (b) Please solve both parts Use the References to access important values if needed for this question. For the reaction
Question 1 part (a)

part (b)
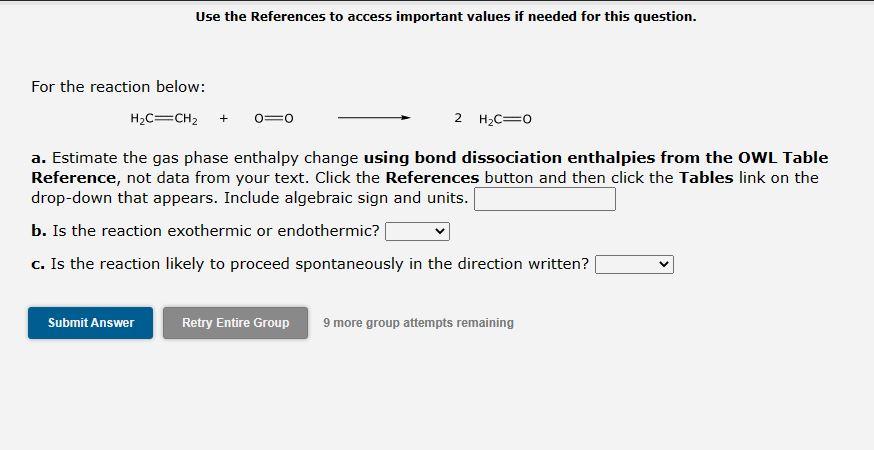
Please solve both parts
Use the References to access important values if needed for this question. For the reaction below: a. Estimate the gas phase enthalpy change using bond dissociation enthalpies from the OWL Table Reference, not data from your text. Click the References button and then click the Tables link on the drop-down that appears. Include algebraic sign and units. b. Is the reaction exothermic or endothermic? C. Is the reaction likely to proceed spontaneously in the direction written? 9 more group attempts remaining Use the References to access important values if needed for this question. For the reaction below: H2C=CH2+O=O2H2C=O a. Estimate the gas phase enthalpy change using bond dissociation enthalpies from the OWL Table Reference, not data from your text. Click the References button and then click the Tables link on the drop-down that appears. Include algebraic sign and units. b. Is the reaction exothermic or endothermic? C. Is the reaction likely to proceed spontaneously in the direction writtenStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


