Answered step by step
Verified Expert Solution
Question
1 Approved Answer
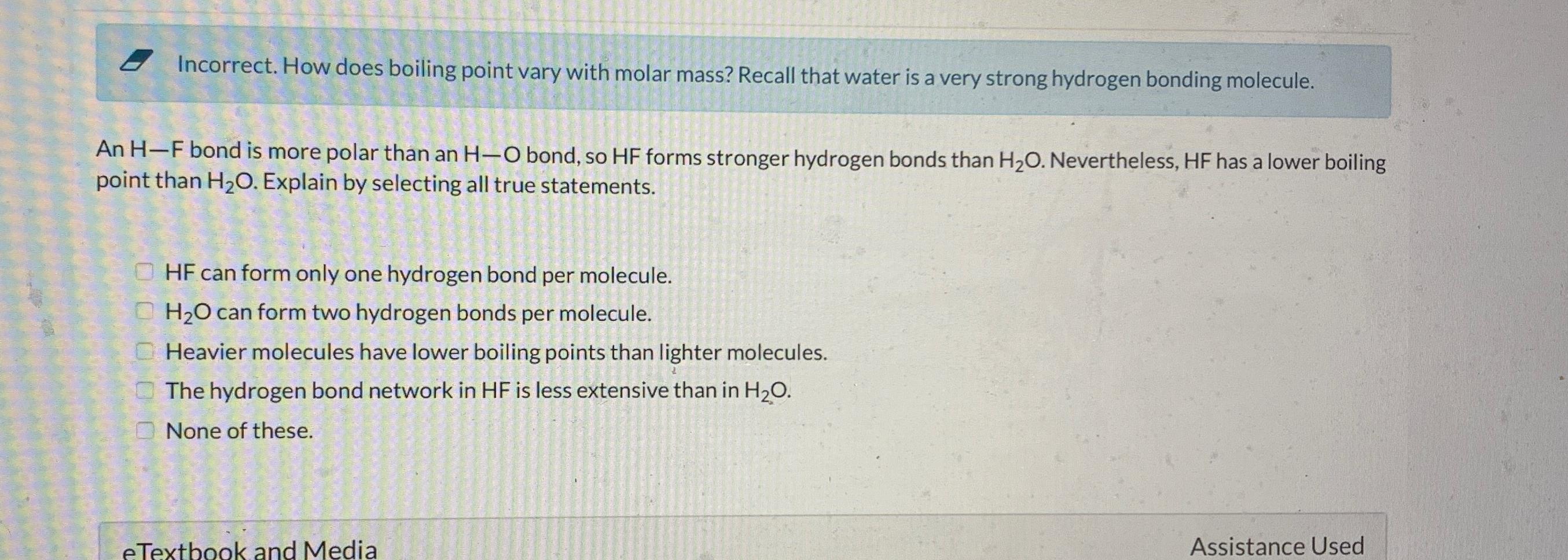
Recall that water is a very strong hydrogen bonding molecule. An H - F bond is more polar than an H - O bond, so
Recall that water is a very strong hydrogen bonding molecule.
An bond is more polar than an bond, so forms stronger hydrogen bonds than Nevertheless, has a lower boiling point than Explain by selecting all true statements.
HF can form only one hydrogen bond per molecule.
can form two hydrogen bonds per molecule.
Heavier molecules have lower boiling points than lighter molecules.
The hydrogen bond network in is less extensive than in
None of these.
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


