Answered step by step
Verified Expert Solution
Question
1 Approved Answer
relating different forms of the equilibrium constant For chemical reactions involving ideal gaises, the equilibrium constant K can be expressed either in terms of the
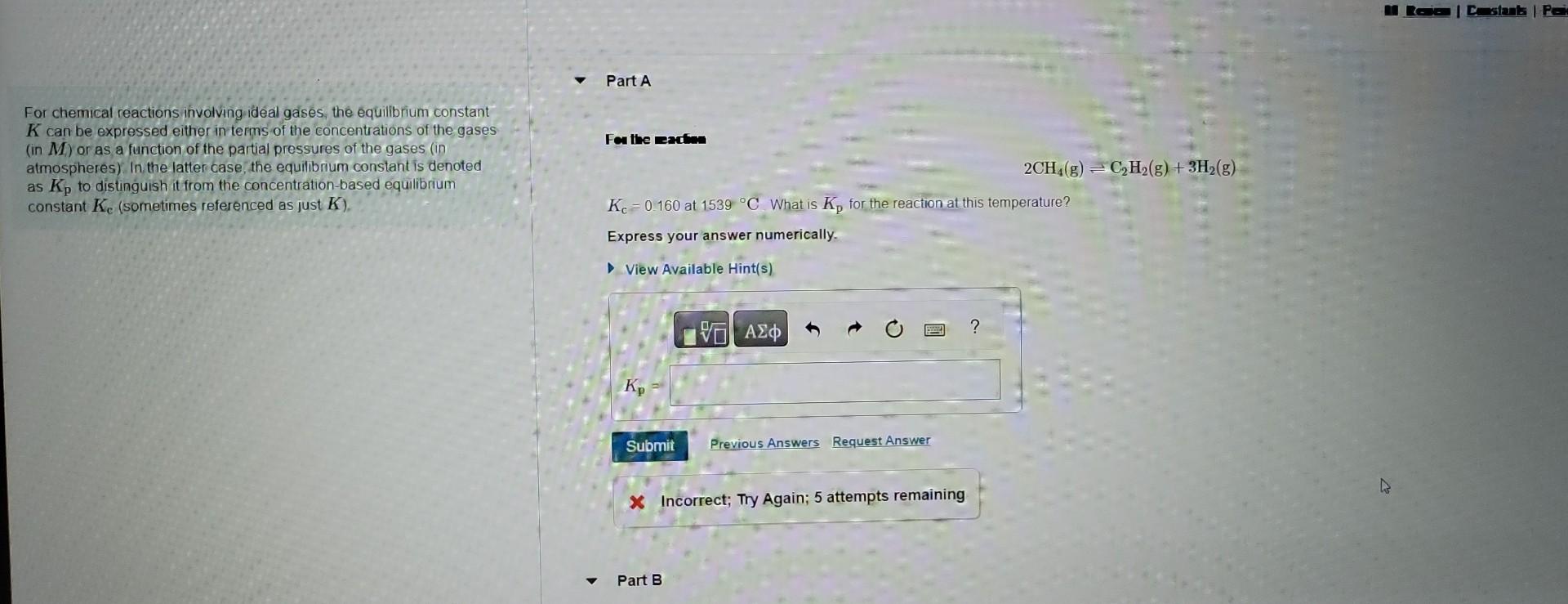
relating different forms of the equilibrium constant
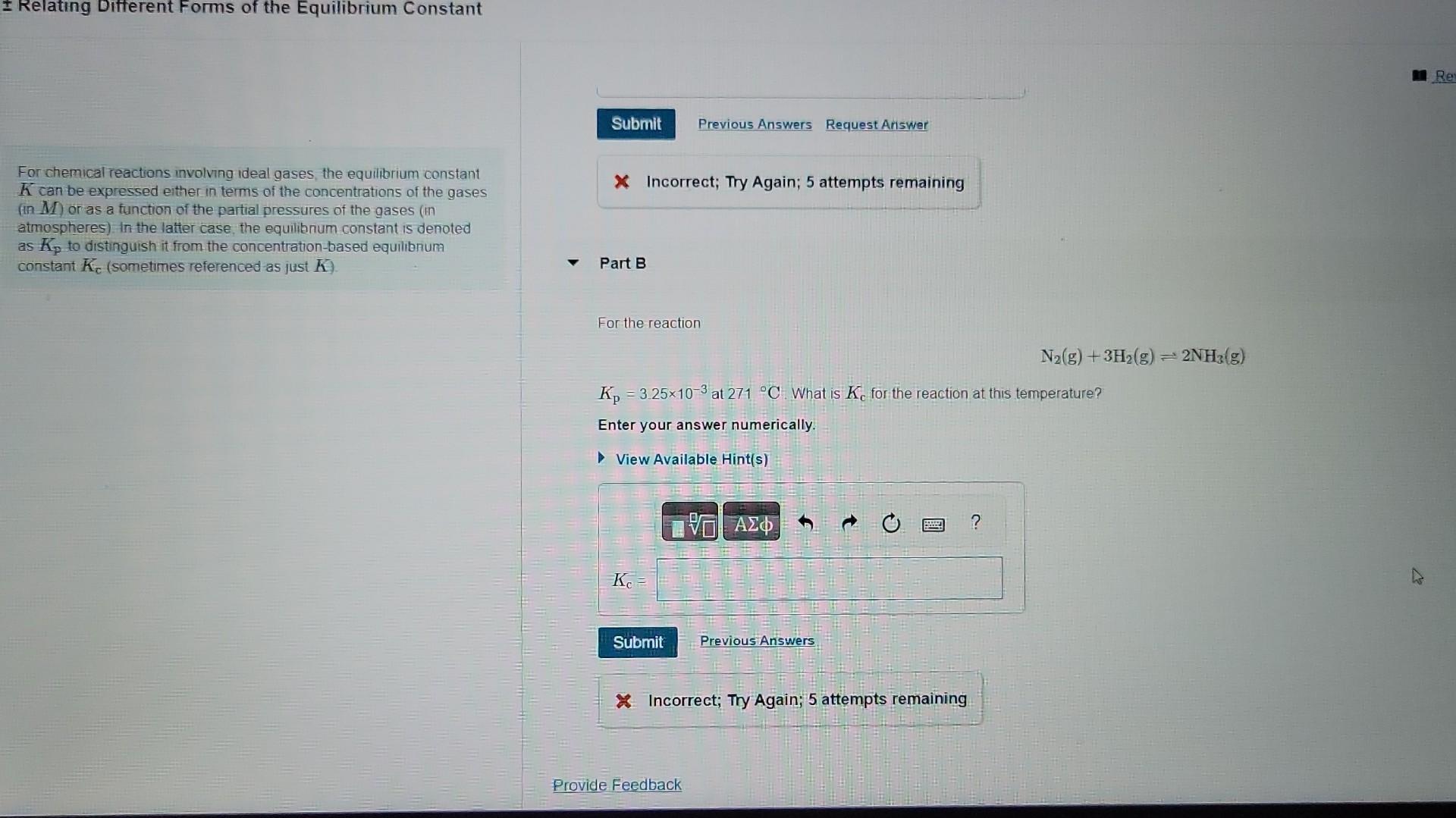
For chemical reactions involving ideal gaises, the equilibrium constant K can be expressed either in terms of the concentrations of the gases (in M ) or as a function of the partial pressures of the gases (in Farthe madia atmospheres) In the latter case, the equilibrium constant is denoted as Kp to distinguish it from the concentration-based equilibrium constant Kc (sometimes referenced as just K ). Kc=0.160 at 1539C. What is Kp for the reaction at this temperature? Express your answer numerically. For chemical reactions involving ideal gases, the equilibrium constant K can be expressed either in terms of the concentrations of the gases x Incorrect; Try Again; 5 attempts remaining (in M ) or as a function of the partial pressures of the gases (in atmospheres). In the latter case, the equilibnum constant is denoted as Kp to distinguish it from the concentration-based equilibnum constant Kc (sometimes referenced as just K ). Part B For the reaction N2(g)+3H2(g)2NH3(g) Kp=3.25103 at 27C. What is Kc for the reaction at this temperature? Enter your answer numerically. In Incorrect; Try Again; 5 attempts remainingStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


