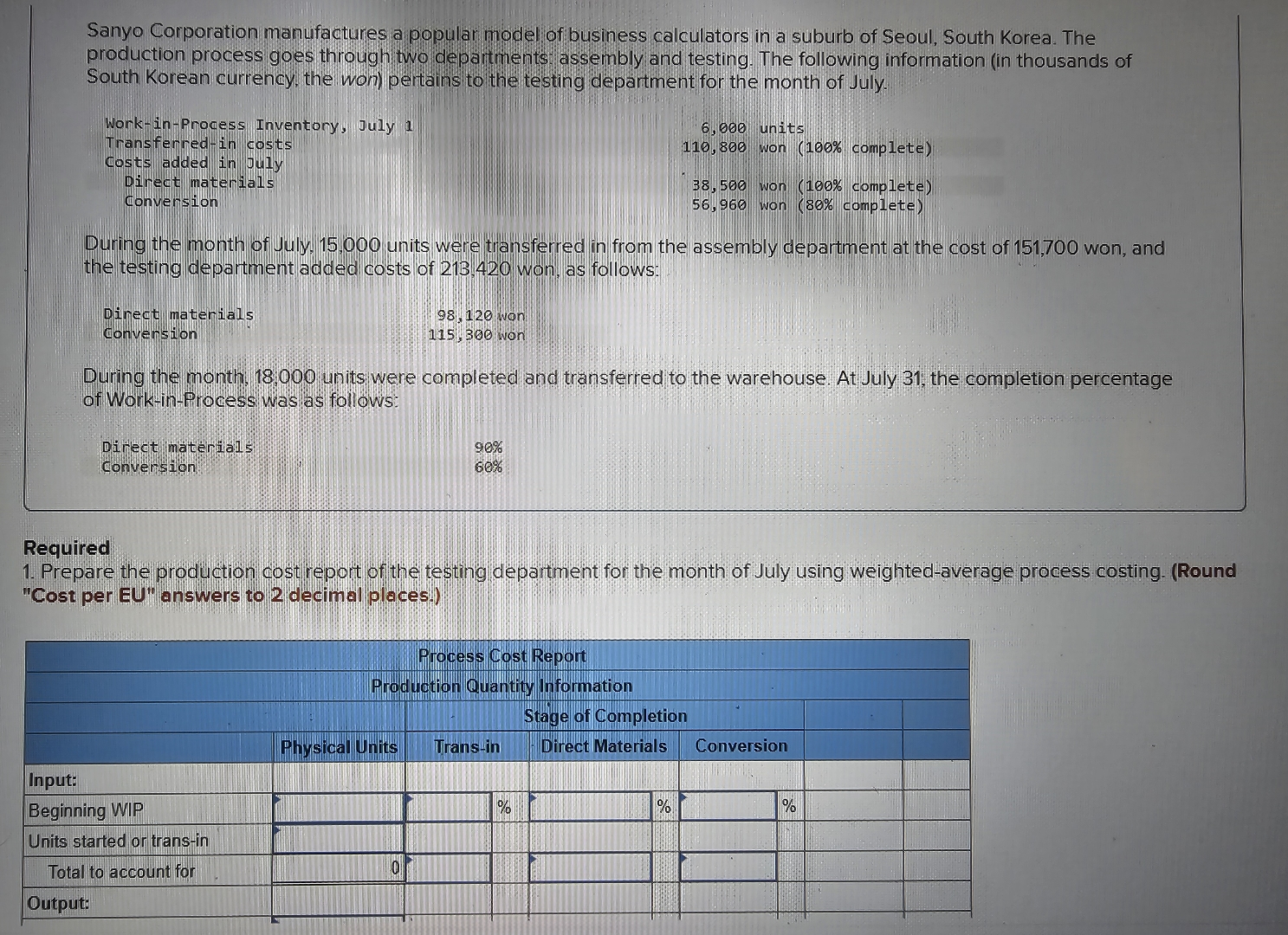
Sanyo Corporation manufactures a popular model of business calculators in a suburb of Seoul, South Korea....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Sanyo Corporation manufactures a popular model of business calculators in a suburb of Seoul, South Korea. The production process goes through two departments assembly and testing. The following information (in thousands of South Korean currency, the won) pertains to the testing department for the month of July. Work-in-Process Inventory, July 1 Transferred-in costs Costs added in July Direct materials Conversion 6,000 units 110,800 won (100% complete) 38,500 won (100% complete) 56,960 won (80% complete) During the month of July. 15.000 units were transferred in from the assembly department at the cost of 151,700 won, and the testing department added costs of 213,420 won, as follows: Direct materials Conversion 98,120 won 115 300 won During the month, 18,000 units were completed and transferred to the warehouse. At July 31, the completion percentage of Work-in-Process was as follows: Direct materials Conversion 90% 60% Required 1. Prepare the production cost report of the testing department for the month of July using weighted-average process costing. (Round "Cost per EU" answers to 2 decimal places.) Process Cost Report Production Quantity Information Input: Beginning WIP Units started or trans-in Stage of Completion Physical Units Trans-in Direct Materials Conversion Total to account for Output: 0 % % % Sanyo Corporation manufactures a popular model of business calculators in a suburb of Seoul, South Korea. The production process goes through two departments assembly and testing. The following information (in thousands of South Korean currency, the won) pertains to the testing department for the month of July. Work-in-Process Inventory, July 1 Transferred-in costs Costs added in July Direct materials Conversion 6,000 units 110,800 won (100% complete) 38,500 won (100% complete) 56,960 won (80% complete) During the month of July. 15.000 units were transferred in from the assembly department at the cost of 151,700 won, and the testing department added costs of 213,420 won, as follows: Direct materials Conversion 98,120 won 115 300 won During the month, 18,000 units were completed and transferred to the warehouse. At July 31, the completion percentage of Work-in-Process was as follows: Direct materials Conversion 90% 60% Required 1. Prepare the production cost report of the testing department for the month of July using weighted-average process costing. (Round "Cost per EU" answers to 2 decimal places.) Process Cost Report Production Quantity Information Input: Beginning WIP Units started or trans-in Stage of Completion Physical Units Trans-in Direct Materials Conversion Total to account for Output: 0 % % %
Expert Answer:

Posted Date:
Students also viewed these accounting questions
-
Sanyo Corporation manufactures a popular model of business calculators in a suburb of Seoul, South Korea. The production process goes through two departments, assembly and testing. The following...
-
The current quoted price of a 13% coupon bond is $110. It pays coupon semi-annually. The next coupon will be paid in 6-days (total number of days in this semi-annual period is 181) and the futures...
-
Unrelated parties Amy, Beth, Carla, and Delta Corporation each own 25 of the 100 outstanding shares of Axle Corporation stock. In a transaction that qualifies as a partial liquidation, Axle...
-
Sams Golden Lab Corp. (SGL) maintains a defined benefit pension plan for its employees. On January 1, 2019, SGL finalized the sale of a business segment to Grettas Great Kittens Ltd. (GGK). Terms of...
-
Given the two-dimensional torus $T^{2}$, parameterized in terms of the two real variables $x^{1} \in[0,2 \pi)$ and $x^{2} \in[0,2 \pi)$, consider its embedding into the threedimensional Euclidean...
-
The B&S Novelty and Craft Shop in Bennington, Vermont, sells a variety of quality handmade items to tourists. B&S will sell 300 hand-carved miniature replicas of a Colonial soldier each year, but the...
-
A solution is made by dissolving 26.3 g of chromium(III)sulfate, Cr2(SO4)3, in enough water to make exactly 500 mL ofsolution. Calculate the concentration (molarity) of Cr2(SO4)3 inmol/L (M). M...
-
Explain the role of physical activation time on the physical and electrochemical properties of supercapacitor electrodes made from betel nut?
-
Read the information from the following excel sheet and fill the required formulas in the cells that are marked with alphabets "a" through "I". 1 2 3 MIS 10 B Amount of Loan Period of Loan Rate of...
-
Design for childcare centres a quality of : - Art Corner -construction corner And what Theories/ policies?
-
How can you apply critical thinking to the topic: GAY MARRIAGE, LIBERALISM, AND RECOGNITION: THE CASE FOR EQUAL TREATMENT. Support your answer. ..
-
5. (20 points) This question will guide you to proving that ax dx 1) ' e = lim (1 + N0 g'(x) = lim Ax0 n Substitute g(x) = e* into this definition = lim(1 + n) n0 = d a) (2 points) First, we need to...
-
Evaluate the following limits. Upload a photo of your work. sin 5x a. lim 1-0 I 2 b. lim x-cos x COS
-
Read the article United States District Court, D. Connecticut. Michelle THIBEAULT, Plaintiff v. SCAP MOTORS, INC. a/k/a Scap Automotive Group, Defendant. And give your response
-
Distinguish between the work performed by public accountants and the work performed by accountants in commerce and industry and in not-for-profit organisations.
-
Calculate the volume occupied by isopropanol vapour at \(200^{\circ} \mathrm{C}\) and 10 bar by using (a) Ideal gas equation of state (b) Virial equation of state (c) Virial equation of state \[...
-
The Dieterici equation of state is given by \[ P(V-b) \exp \left(\frac{a}{R T V} ight)=R T \] where \(a\) and \(b\) are constants. Develop the relations to determine the constants \(a\) and \(b\) in...
-
At \(17^{\circ} \mathrm{C}\), at constant pressure, the heat of combustion of amorphous carbon is 96960 cal and that of \(\mathrm{CO}\) to \(\mathrm{CO}_{2}\) is \(67960 \mathrm{cal}\). Determine the...

Study smarter with the SolutionInn App

