Answered step by step
Verified Expert Solution
Question
1 Approved Answer
solve and show work In an aqueous chloride solution cobalt(II) exists in equilibrium with the complex ion CoCl42Co2+(aq) is pink and CoCl42(aq) is blue. At
solve and show work
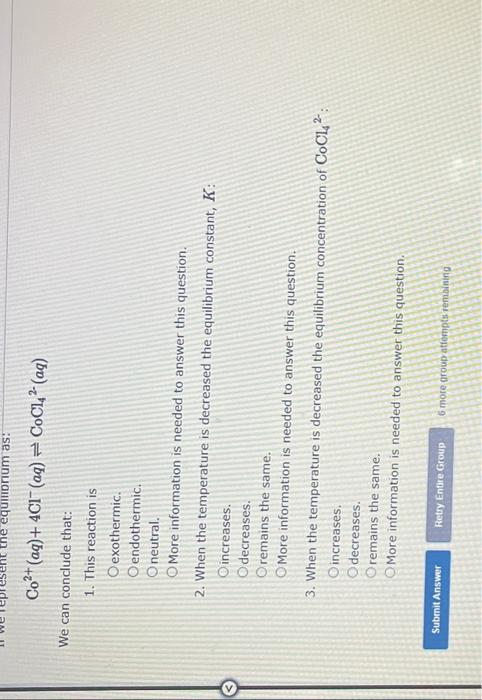
In an aqueous chloride solution cobalt(II) exists in equilibrium with the complex ion CoCl42Co2+(aq) is pink and CoCl42(aq) is blue. At low temperature the pink color predominates. At high temperature the blue color is strong. If we represent the equilibrium as: Co2+(aq)+4Cl(aq)CoCl42(aq) We can conclude that: 1. This reaction is exothermic. endothermic. neutral. More information is needed to answer this question. 2. When the temperature is decreased the equilibrium constant, K : increases. decreases. remains the same. Co2+(aq)+4Cl(aq)CoCl42(aq) conclude that: 1. This reaction is exothermic. endothermic. neutral. More information is needed to answer this question. 2. When the temperature is decreased the equilibrium constant, I increases. decreases. remains the same. More information is needed to answer this question. 3. When the temperature is decreased the equilibrium concentratio increases. decreases. remains the same. More information is needed to answer this 

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


