Answered step by step
Verified Expert Solution
Question
1 Approved Answer
solve it How many grams of NaCl are required to precipitate most of the Ag+ ions from 5.50 x102 mL of 0.0487 M AgNO3 solution?
solve it 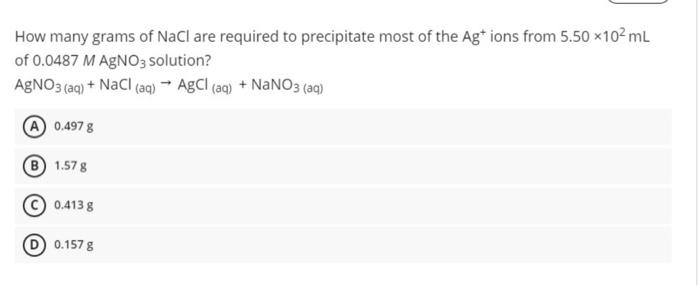


How many grams of NaCl are required to precipitate most of the Ag+ ions from 5.50 x102 mL of 0.0487 M AgNO3 solution? AgNO3(aq) + NaCl (aq) AgCl(aq) + NaNO3 (ag) A 0.4978 1.578 0.4138 0.157 g A 249 mL benzene solution containing 2.89 g of an organic polymer has an osmotic pressure of 7.59 mm Hg at 11C. Calculate the molar mass of the polymer.[R=0.0821 L atm/K mol] A 2.13 X 104 g/mol B 3.30 X 109 g/mol 2.71 X 104 g/mol 1.79 X 104 g/mol What is the solubility of O2(8) in water at 25C if the atmospheric pressure is 841 mmHg? The Henry's law constant for oxygen gas at 25C is 1.3 x 10-3 mol/L.am 1.4 X10-3M B 2.7 X 10 -4 M 9.6 X10 -3M 6.2 X10-3 M 


Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


