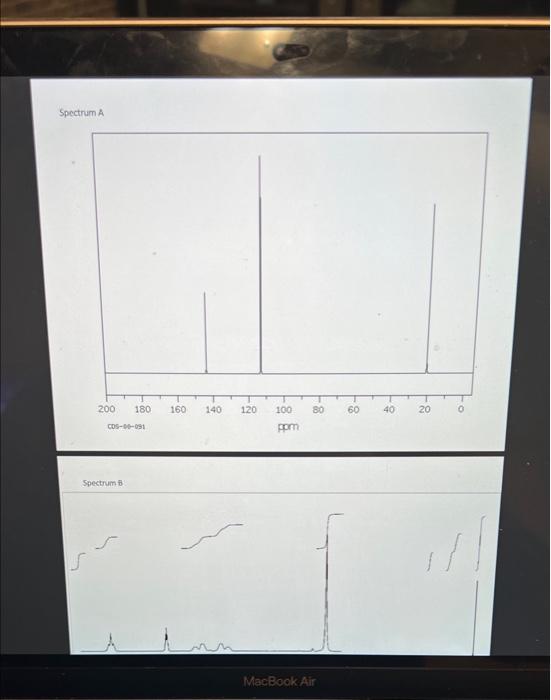
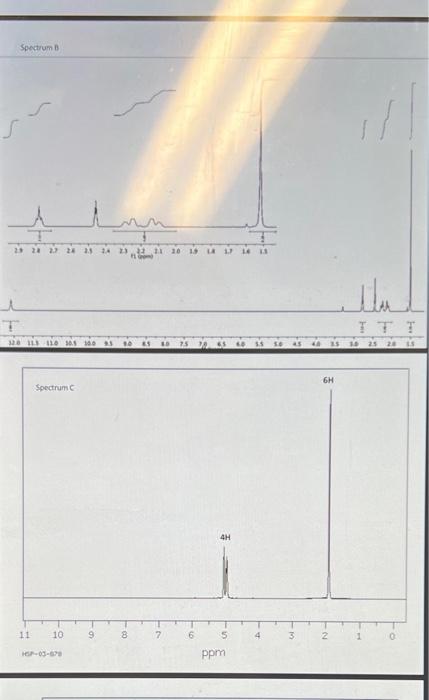
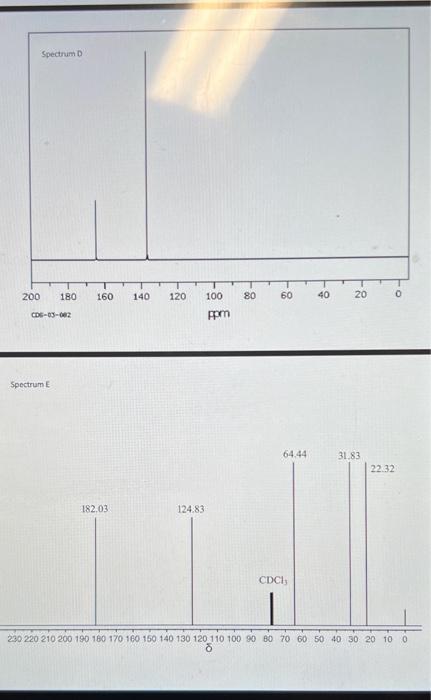
Spectrum A Spectrum of Spectrum D cos63622 pprn Spectrum E One of the methods of chemical characterization that you are discussing in lecture used to obtain magnetic resonance spectroscopy, or NMR. This instrumental ('H-NMR) or carbon atoms ( 3CNMR) in a information about the numbers and types of some of the starting materials and products of this week's molecule. 'H-NMR and 'Sale Canvas: maleic anhydride, 2,3-dimethyl-1,3-butadiene, the Diels-Alder product. reaction are available in fred by hydrolysis of the Diels-Alder product. The spectra are not labelled with and the diacid product founds. It is your task to use the new knowledge you are gaining concerning the corresponding spectra with the correct structures. For each spectrum: a. draw clearly the structure that produced that spectrum... (2.5 points total/ 0.5 points per spectrum) b. ...and provide a couple of sentences explaining each choice. For each spectrum, explain (1) the number of signals in the spectrum, (2) their chemical shifts in ppm, and (3) the splitting of each signal. (1.25 points total/ 0.25 points per spectrum) NOTE: Your discussion must conclusively demonstrate why the compound matches the spectrum to gain the 0.25 points for that spectrum. Separate your discussion into sections by spectrum with a heading of the form "Spectrum X" each section. c. Answer the following question for Spectrum D only. What functional group produces the signal with chemical shift between160-170 ppm? ( 0.25 points) Spectrum A Spectrum of Spectrum D cos63622 pprn Spectrum E One of the methods of chemical characterization that you are discussing in lecture used to obtain magnetic resonance spectroscopy, or NMR. This instrumental ('H-NMR) or carbon atoms ( 3CNMR) in a information about the numbers and types of some of the starting materials and products of this week's molecule. 'H-NMR and 'Sale Canvas: maleic anhydride, 2,3-dimethyl-1,3-butadiene, the Diels-Alder product. reaction are available in fred by hydrolysis of the Diels-Alder product. The spectra are not labelled with and the diacid product founds. It is your task to use the new knowledge you are gaining concerning the corresponding spectra with the correct structures. For each spectrum: a. draw clearly the structure that produced that spectrum... (2.5 points total/ 0.5 points per spectrum) b. ...and provide a couple of sentences explaining each choice. For each spectrum, explain (1) the number of signals in the spectrum, (2) their chemical shifts in ppm, and (3) the splitting of each signal. (1.25 points total/ 0.25 points per spectrum) NOTE: Your discussion must conclusively demonstrate why the compound matches the spectrum to gain the 0.25 points for that spectrum. Separate your discussion into sections by spectrum with a heading of the form "Spectrum X" each section. c. Answer the following question for Spectrum D only. What functional group produces the signal with chemical shift between160-170 ppm? ( 0.25 points)