Question
Table A-11 (page 872) reports KH=4.01x10+4 atm for O2 at 20C. These KH values use gas phase partial pressure in atmospheres and aqueous phase concentration
Table A-11 (page 872) reports KH=4.01x10+4 atm for O2 at 20C. These KH values use gas phase partial pressure in atmospheres and aqueous phase concentration in mole fraction (which is dimensionless) and are often indicated with the symbol K. Convert this K to KH in M/atm. Assume the concentration of O2 in water is dilute, meaning the number of moles of O2 in a sample is small compared to the number of moles of water and that the density is negligibly different from the density of water (1.00 kg/L). What saturation O2 concentration is predicted in mg/L? How does this compare to the value in Table A-2 at 20C? Use molar masses 32.00 and 18.02 for O2 and H2O.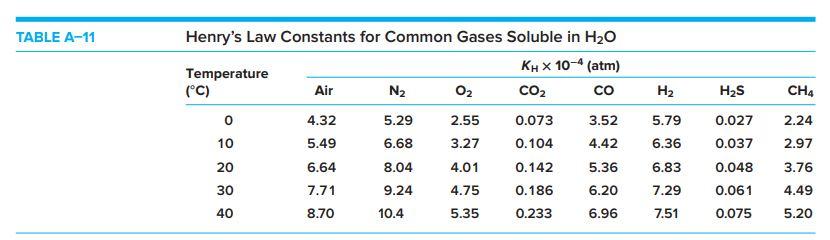
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


