Answered step by step
Verified Expert Solution
Question
1 Approved Answer
thank you for your help!!! there were 8 reactions for part b, with vsrying amounts of water and H2O2 to bring all solutions to 11mL
thank you for your help!!! 

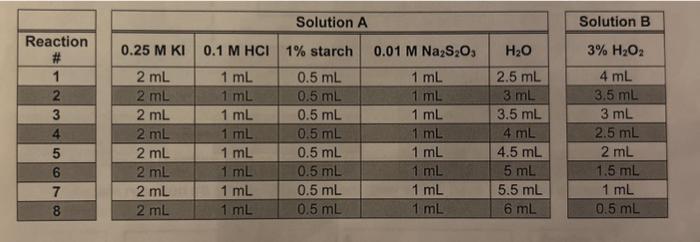
there were 8 reactions for part b, with vsrying amounts of water and H2O2 to bring all solutions to 11mL
1. The instructor prepared a solution of Na2S2O3 which she thought was 0.1M, but it turned out to be 0.01M. How would that affect the rate of the reaction in part B. Would the rate be faster or slower? Explain. \begin{tabular}{|c|c|c|c|c|} \hline \multicolumn{5}{|c|}{ Solution A } \\ \hline \hline 0.25MKI & 0.1MHCl & 1% starch & 0.01MNa2S2O3 & H2O \\ \hline 2mL & 1mL & 0.5mL & 1mL & 2.5mL \\ \hline 2mL & 1mL & 0.5mL & 1mL & 3mL \\ \hline 2mL & 1mL & 0.5mL & 1mL & 3.5mL \\ \hline 2mL & 1mL & 0.5mL & 1mL & 4mL \\ \hline 2mL & 1mL & 0.5mL & 1mL & 4.5mL \\ \hline 2mL & 1mL & 0.5mL & 1mL & 5mL \\ \hline 2mL & 1mL & 0.5mL & 1mL & 5.5mL \\ \hline 2mL & 1mL & 0.5mL & 1mL & 6mL \\ \hline \end{tabular} \begin{tabular}{|c|} \hline Solution B \\ \hline 3%H2O2 \\ \hline 4mL \\ \hline 3.5mL \\ \hline 3mL \\ \hline 2.5mL \\ \hline 2mL \\ \hline 1.5mL \\ \hline 1mL \\ \hline 0.5mL \\ \hline \end{tabular} Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


