Question
The development of a photographic image on film is a process controlled by kinetics. Exposed silver halide crystals in the film are reduced to silver
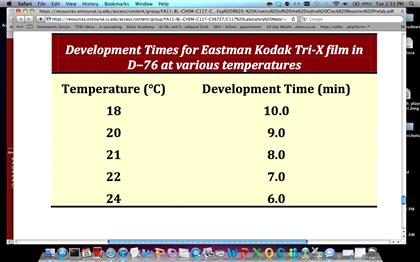
The development of a photographic image on film is a process controlled by kinetics. Exposed silver halide crystals in the film are reduced to silver in a developing solution. The development process is performed for a certain amount of time, which depends on the temperature of the developing solution, and with agitation to ensure transfer of reagents to the film. The time required for development at a particular temperature is inversely proportional to the rate constant for the process (in other words, k = 1/Development Time). The table below gives the development times (in minutes) for Eastman Kodak’s Tri‐X film in D‐76 developer. Use the Arrhenius equation to determine the activation energy (Ea)
in kJ mol for the process. Attach any plots to this prelab and clearly show all work.
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


