Question
The following reaction between ethyl alcohol and ethyl bromide forms diethyl ether via a substitution reaction (followed by deprotonation). (a) Determine the electrophilic site
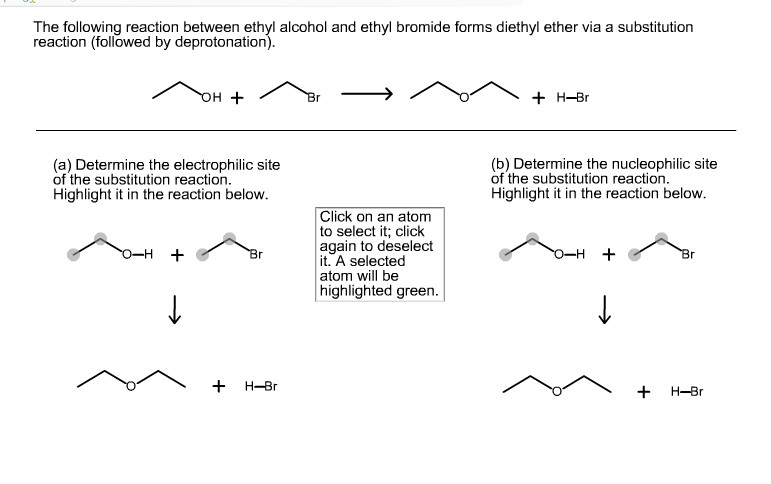
The following reaction between ethyl alcohol and ethyl bromide forms diethyl ether via a substitution reaction (followed by deprotonation). (a) Determine the electrophilic site of the substitution reaction. Highlight it in the reaction below. O-H + OH + + Br H-Br Br Click on an atom to select it; click again to deselect it. A selected atom will be highlighted green. + H-Br (b) Determine the nucleophilic site of the substitution reaction. Highlight it in the reaction below. O-H + + H-Br
Step by Step Solution
There are 3 Steps involved in it
Step: 1
General Guidance Electrophile is an electron loving and nucleophile is a nucleus loving Electron pai...
Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get StartedRecommended Textbook for
Organic Chemistry
Authors: L. G. Wade Jr.
8th edition
321768418, 978-0321768414
Students also viewed these Chemistry questions
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
View Answer in SolutionInn App



