Answered step by step
Verified Expert Solution
Question
1 Approved Answer
The following reaction has an equilibrium constant of Kp= 2.26 x 10^4 at 298 K: CO(g)+2H2(g)CH3OH(g) Part A: calculate K'p for the following reaction: CH3OH(g)
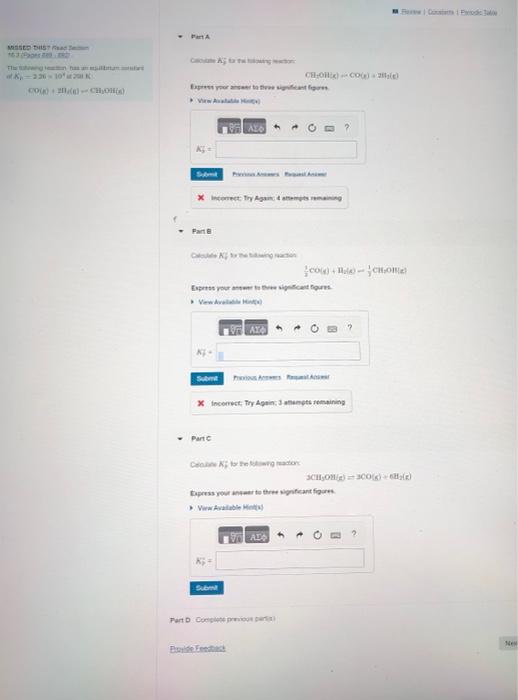
The following reaction has an equilibrium constant of Kp= 2.26 x 10^4 at 298 K: 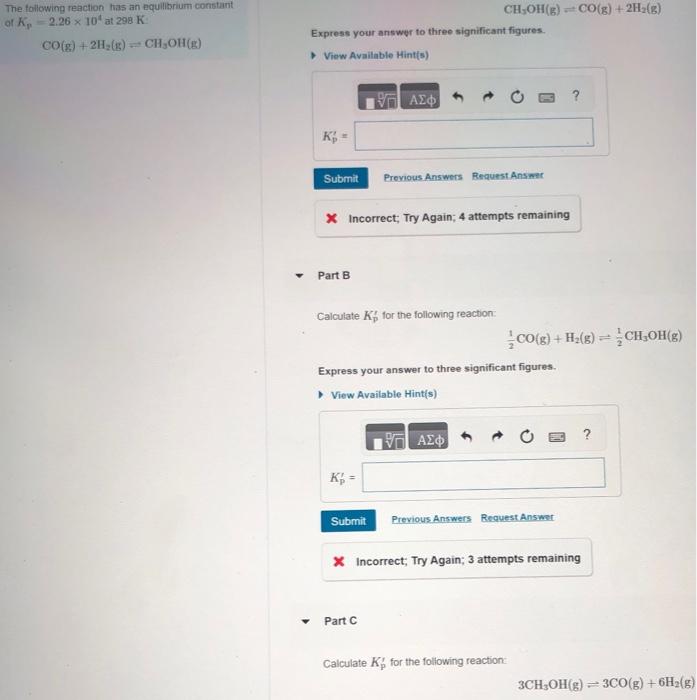
ofkn=228=104if2aiKi. X intwewet, Try Againci amemuts masining Part E 31co(4)+1B()y1cutorMa) X. incervect. Try A gitis; 3 amemets rempinitg Parte schigovia)=3C0(s)+6ta(e) tapress your khsear to three slguficant figures. The following reaction has an equilibrium constant ofKp=2.26104at298KCO(g)+2H2(g)=CH3OH(g) Express your answer to three significant figures. x Incorrect; Try Again; 4 attempts remaining Part B Calculate Kp for the following reaction: 21CO(g)+H2(g)21CH3OH(g) Express your answer to three significant figures. x Incorrect; Try Again; 3 attempts remaining Part C Calculate Kp for the following reaction: 3CH3OH(g)3CO(g)+6H2(g) CO(g)+2H2(g)CH3OH(g)
Part A: calculate K'p for the following reaction: CH3OH(g) CO(g) + 2H2(g)
Part B: calculate K'p for the following reaction:
1/2CO(g)+H2(g) 1/2 CH3OH(g)
Part C: calculate K'p for the following reaction:
3 CH3OH(g) 3CO(g)+6H2(g)

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


