Answered step by step
Verified Expert Solution
Question
1 Approved Answer
The gas phase reaction between methanol ( C H 3 O H ) and acetic acid ( C H 3 C O O H )
The gas phase reaction between methanol and acetic acid to form methyl acetate and water
takes place in a continuous reactor fed with equimolar quantities of methanol and acetic acid. If the equilibrium constant for this reaction equals determine the conversion of methanol in the reactor assuming the exit gases are at equilibrium.
PLEASE SEE: Please work it out correctly and send the answer written down on a paper. Thanks!
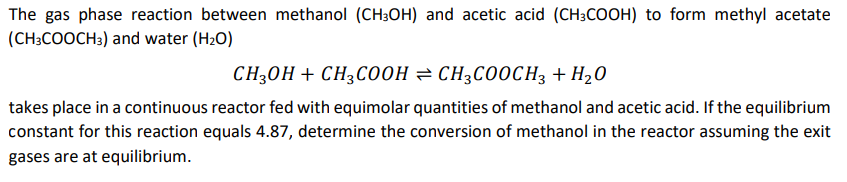
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


