Answered step by step
Verified Expert Solution
Question
1 Approved Answer
The Solubility Product Constant for lead carbonate is 1.51013. The molar solubility of lead carbonate in a 0.293M lead nitrate solution is The Solubility Product
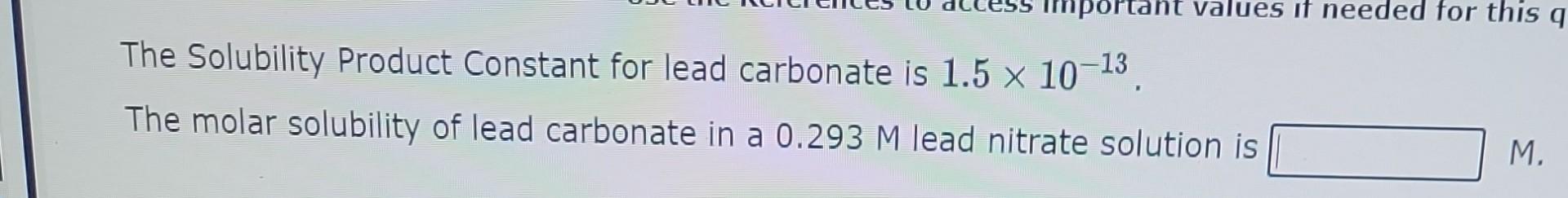

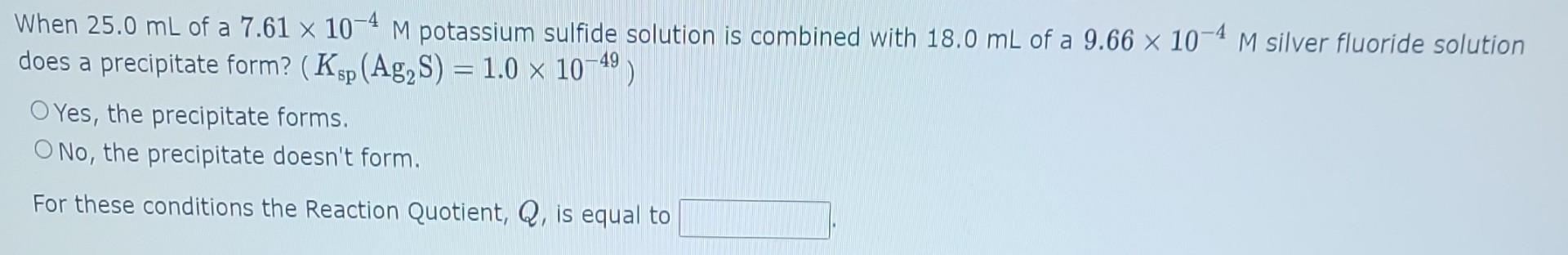

The Solubility Product Constant for lead carbonate is 1.51013. The molar solubility of lead carbonate in a 0.293M lead nitrate solution is The Solubility Product Constant for lead fluoride is 3.7108. The molar solubility of lead fluoride in a 0.179M sodium fluoride solution is When 25.0mL of a 7.61104M potassium sulfide solution is combined with 18.0mL of a 9.66104M silver fluoride solution does a precipitate form? (Ksp(Ag2S)=1.01049) Yes, the precipitate forms. No, the precipitate doesn't form. For these conditions the Reaction Quotient, Q, is equal to When 18.0mL of a 4.00104M potassium hydroxide solution is combined with 25.0mL of a 9.71104M copper(II) nitrate solution does a precipitate form? (Ksp(Cu(OH)2)=1.61019) Yes, the precipitate forms. No, the precipitate doesn't form. For these conditions the Reaction Quotient, Q, is equal to
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


