Answered step by step
Verified Expert Solution
Question
1 Approved Answer
The standard reduction potential of Cr3+ and Ni2+ have been experimentally determined as 0.732V and 0.243V respectively. i. Complete the following sentence by entering numbers
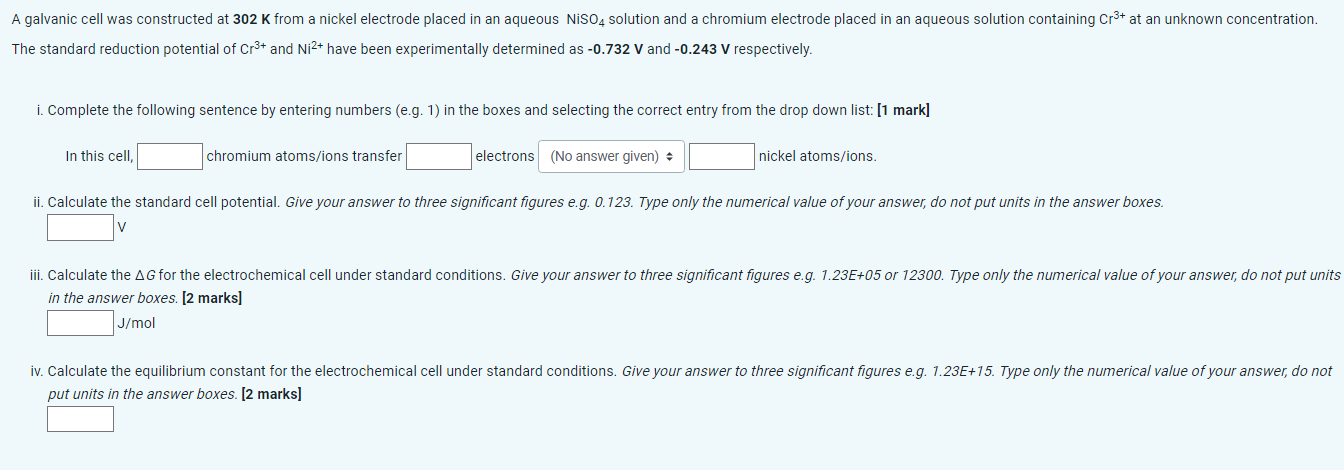 The standard reduction potential of Cr3+ and Ni2+ have been experimentally determined as 0.732V and 0.243V respectively. i. Complete the following sentence by entering numbers (e.g. 1) in the boxes and selecting the correct entry from the drop down list: [1 mark] In this cell, chromium atoms/ions transfer electrons nickel atoms/ions. in the answer boxes. [2 marks] J/mol put units in the answer boxes. [2 marks] The standard reduction potential of Cr3+ and Ni2+ have been experimentally determined as 0.732V and 0.243V respectively. i. Complete the following sentence by entering numbers (e.g. 1) in the boxes and selecting the correct entry from the drop down list: [1 mark] In this cell, chromium atoms/ions transfer electrons nickel atoms/ions. in the answer boxes. [2 marks] J/mol put units in the answer boxes. [2 marks]
The standard reduction potential of Cr3+ and Ni2+ have been experimentally determined as 0.732V and 0.243V respectively. i. Complete the following sentence by entering numbers (e.g. 1) in the boxes and selecting the correct entry from the drop down list: [1 mark] In this cell, chromium atoms/ions transfer electrons nickel atoms/ions. in the answer boxes. [2 marks] J/mol put units in the answer boxes. [2 marks] The standard reduction potential of Cr3+ and Ni2+ have been experimentally determined as 0.732V and 0.243V respectively. i. Complete the following sentence by entering numbers (e.g. 1) in the boxes and selecting the correct entry from the drop down list: [1 mark] In this cell, chromium atoms/ions transfer electrons nickel atoms/ions. in the answer boxes. [2 marks] J/mol put units in the answer boxes. [2 marks] Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


