Answered step by step
Verified Expert Solution
Question
1 Approved Answer
This is all one question, thank you in advance Kinetics 1. The rate of the reaction NO2+CONO+CO2 depends only on the concentration of nitrogen dioxide
This is all one question, thank you in advance 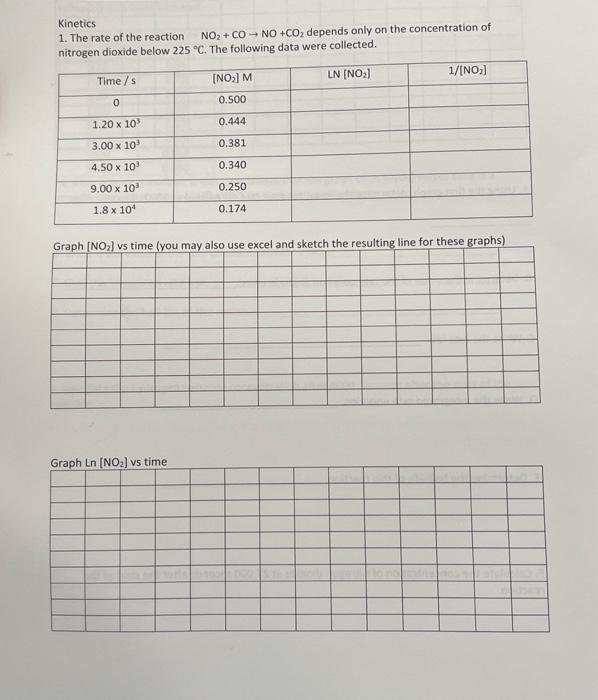
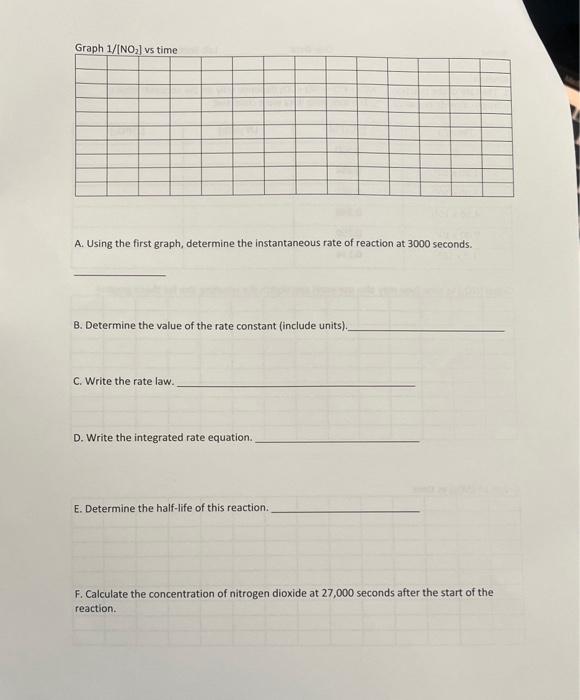
Kinetics 1. The rate of the reaction NO2+CONO+CO2 depends only on the concentration of nitrogen dioxide below 225C. The following data were collected. Feanh fMn l we time funu mav alcn usp pxcal and sketch the resulting line for these graphs) A. Using the first graph, determine the instantaneous rate of reaction at 3000 seconds. B. Determine the value of the rate constant (include units). c. Write the rate law. D. Write the integrated rate equation. E. Determine the half-life of this reaction. F. Calculate the concentration of nitrogen dioxide at 27,000 seconds after the start of the reaction. Kinetics 1. The rate of the reaction NO2+CONO+CO2 depends only on the concentration of nitrogen dioxide below 225C. The following data were collected. Feanh fMn l we time funu mav alcn usp pxcal and sketch the resulting line for these graphs) A. Using the first graph, determine the instantaneous rate of reaction at 3000 seconds. B. Determine the value of the rate constant (include units). c. Write the rate law. D. Write the integrated rate equation. E. Determine the half-life of this reaction. F. Calculate the concentration of nitrogen dioxide at 27,000 seconds after the start of the reaction 

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


