Answered step by step
Verified Expert Solution
Question
1 Approved Answer
This is for the course Reaction Engineering which concerns the design of reactors. using the problem below and the solution, EXPLAIN THE GIVEN SOLUTION AND
This is for the course Reaction Engineering which concerns the design of reactors. using the problem below and the solution, EXPLAIN THE GIVEN SOLUTION AND EXPLAIN WHAT THE PROBLEM SHOULD TEACH CHEMICAL ENGINEERING STUDENTS.
Problem: The first order gas phase reaction A B is taking place in a constant volume batch reactor. The initial pressure, which is constituted with A and the rest inerts is atm. If the rate constant for the reaction is min how much time would be needed to reach a pressure of atm in the reactor.
Solution:
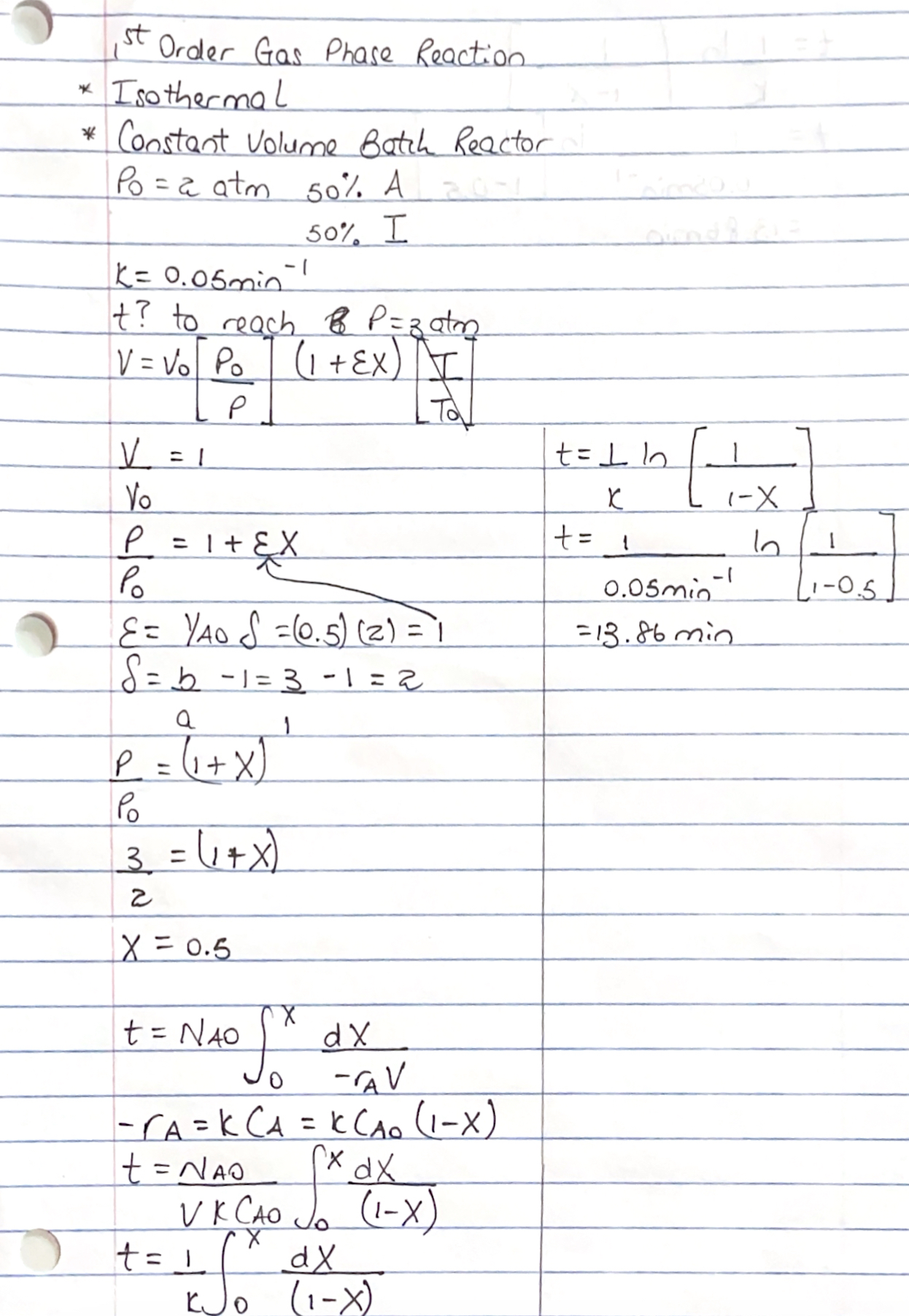
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


