Answered step by step
Verified Expert Solution
Question
1 Approved Answer
This question has 4 parts (A,B,C,D). Please answer each one in tge form of a ratio This is all of the info they offer. Nothing
This question has 4 parts (A,B,C,D). Please answer each one in tge form of a ratio 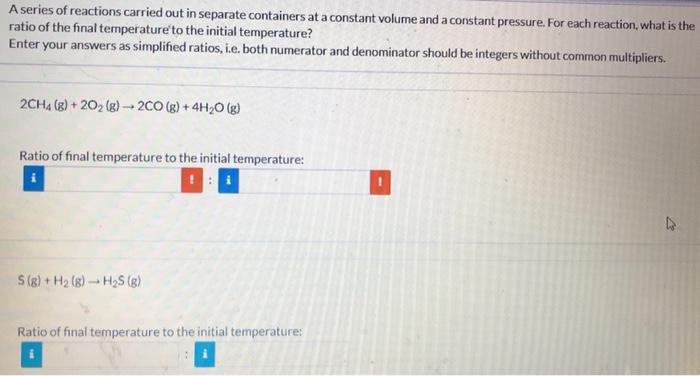


This is all of the info they offer. Nothing is missing. 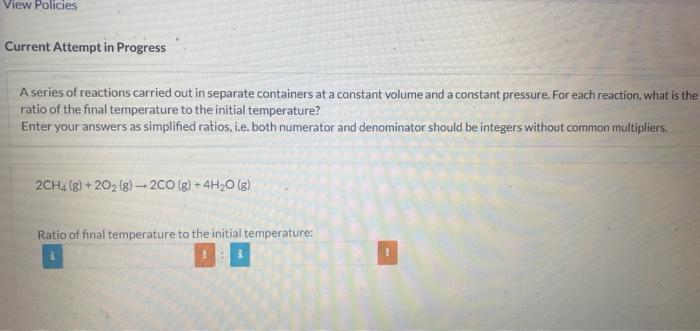

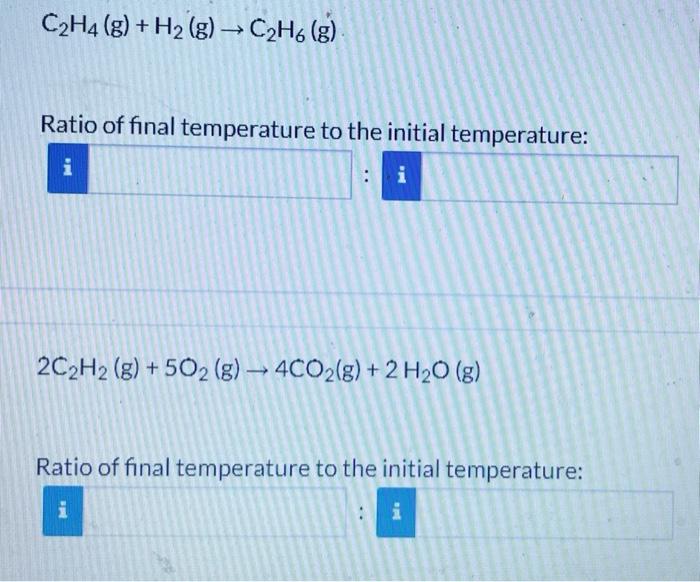
A series of reactions carried out in separate containers at a constant volume and a constant pressure. For each reaction, what is the ratio of the final temperature to the initial temperature? Enter your answers as simplified ratios, i.e. both numerator and denominator should be integers without common multipliers. 2CH4(g)+2O2(g)2CO(g)+4H2O(g) Ratio of final temperature to the initial temperature: S(g)+H2(g)H2S(g) Ratio of final temperature to the initial temperature: C2H4(g)+H2(g)C2H6(g) Ratio of final temperature to the initial temperature: 2C2H2(g)+5O2(g)4CO2(g)+2H2O(g) Ratio of final temperature to the initial temperature: A series of reactions carried out in separate containers at a constant volume and a constant pressure. For each reaction, what is the ratio of the final temperature to the initial temperature? Enter your answers as simplified ratios, i.e. both numerator and denominator should be integers without common multipliers. 2CH4(g)+2O2(g)2CO(g)+4H2O(g) S(g)+H2(g)H2S(g) Ratio of final temperature to the initial temperature: C2H4(g)+H2(g)C2H6(g) Ratio of final temperature to the initial temperature: 2C2H2(g)+5O2(g)4CO2(g)+2H2O(g) Ratio of final temperature to the initial temperature: A series of reactions carried out in separate containers at a constant volume and a constant pressure. For each reaction, what is the ratio of the final temperature to the initial temperature? Enter your answers as simplified ratios, i.e. both numerator and denominator should be integers without common multipliers. 2CH4(g)+2O2(g)2CO(g)+4H2O(g) Ratio of final temperature to the initial temperature: S(g)+H2(g)H2S(g) Ratio of final temperature to the initial temperature: C2H4(g)+H2(g)C2H6(g) Ratio of final temperature to the initial temperature: 2C2H2(g)+5O2(g)4CO2(g)+2H2O(g) Ratio of final temperature to the initial temperature: A series of reactions carried out in separate containers at a constant volume and a constant pressure. For each reaction, what is the ratio of the final temperature to the initial temperature? Enter your answers as simplified ratios, i.e. both numerator and denominator should be integers without common multipliers. 2CH4(g)+2O2(g)2CO(g)+4H2O(g) S(g)+H2(g)H2S(g) Ratio of final temperature to the initial temperature: C2H4(g)+H2(g)C2H6(g) Ratio of final temperature to the initial temperature: 2C2H2(g)+5O2(g)4CO2(g)+2H2O(g) Ratio of final temperature to the initial temperature 

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


