Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Use the following data to answer the items below: mass of unknown nonelectrolyte = 1 614 g freezing point of pure solvent = 0.05C mass
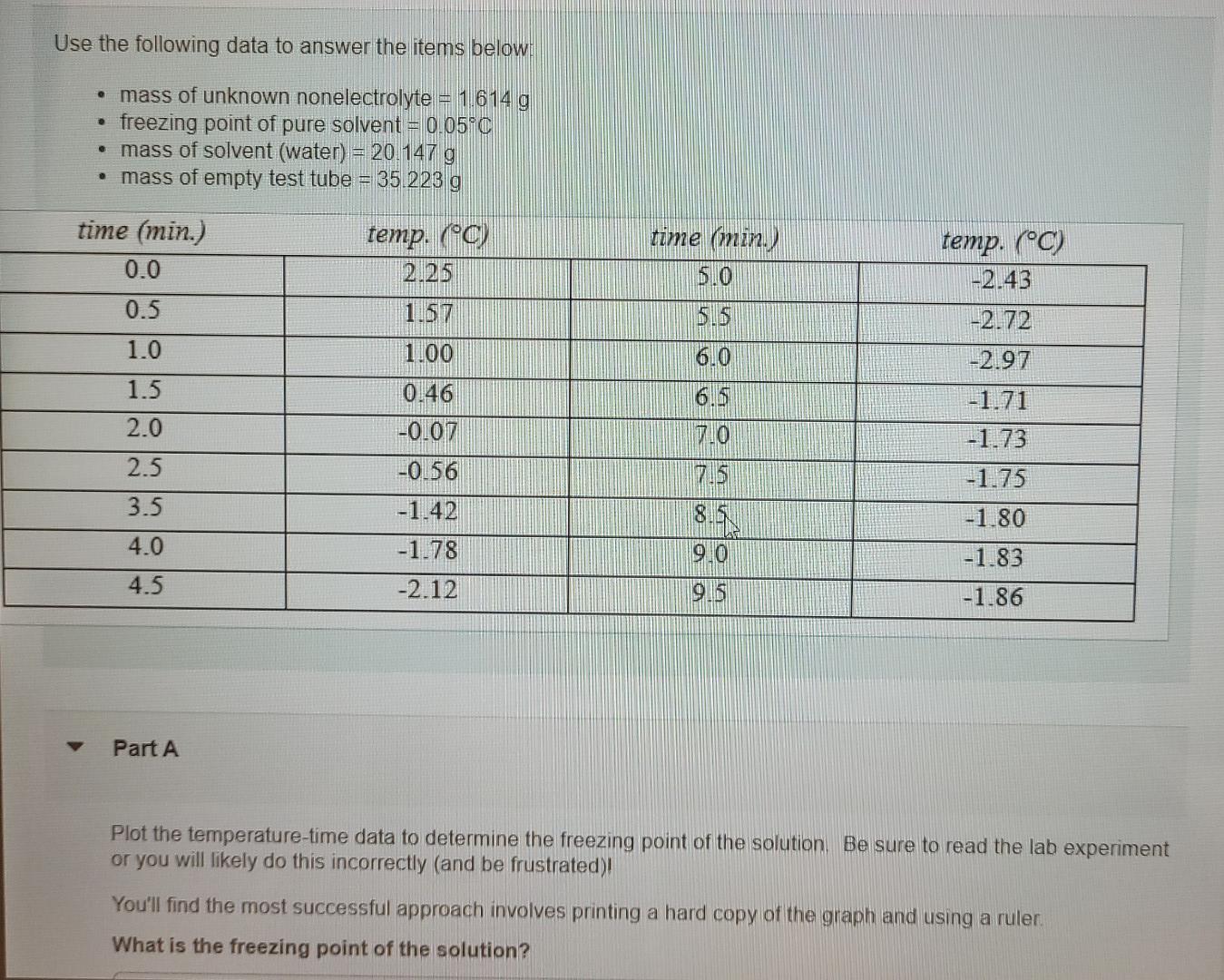
Use the following data to answer the items below: mass of unknown nonelectrolyte = 1 614 g freezing point of pure solvent = 0.05C mass of solvent (water) = 20.147 g mass of empty test tube = 35.223 g time (min.) 8.0 5.5 6.0 time (min.) 0.0 0.5 1.0 1.5 2.0 2.5 3.5 4.0 temp. (0) 2.25 1.57 1.00 0.46 -0.07 -0.56 -1.42 -1.78 -2.12 temp. (C) -2.43 -2.72 -2.97 -1.71 -1.73 -1.75 -1.80 7.0 TIB Ou 9.0 -1.83 4.5 OS -1.86 Part A Plot the temperature-time data to determine the freezing point of the solution. Be sure to read the lab experiment or you will likely do this incorrectly (and be frustrated)! You'll find the most successful approach involves printing a hard copy of the graph and using a ruler. What is the freezing point of the solution
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


