Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Using MATLAB, given the following information: The ideal gas law: and Van der Waals modification of the ideal gas law: Using MATLAB, provide the lines
Using MATLAB, given the following information:
The ideal gas law:
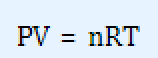
and Van der Waals modification of the ideal gas law:
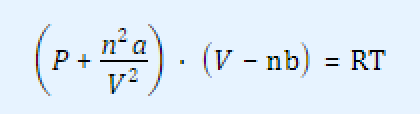
Using MATLAB, provide the lines of code to do the following calculations:
a) Use both the ideal gas law and vad der Waals' equation to calculate the temperature of water vapor (steam), given the following data.

b) Leaving the other variables the same as (a), find the value of temperature (T) for 10 values of pressure from 0 bar to 400 bar - use both the ideal gas law and Van der Waals
PV = nRT
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


