Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Using the data and the diagram provided below, determine the enthalpies of solution of sodium chloride Enthalpy of lattice formation NaCl=800kJmol1 Enthalpy of hydration of
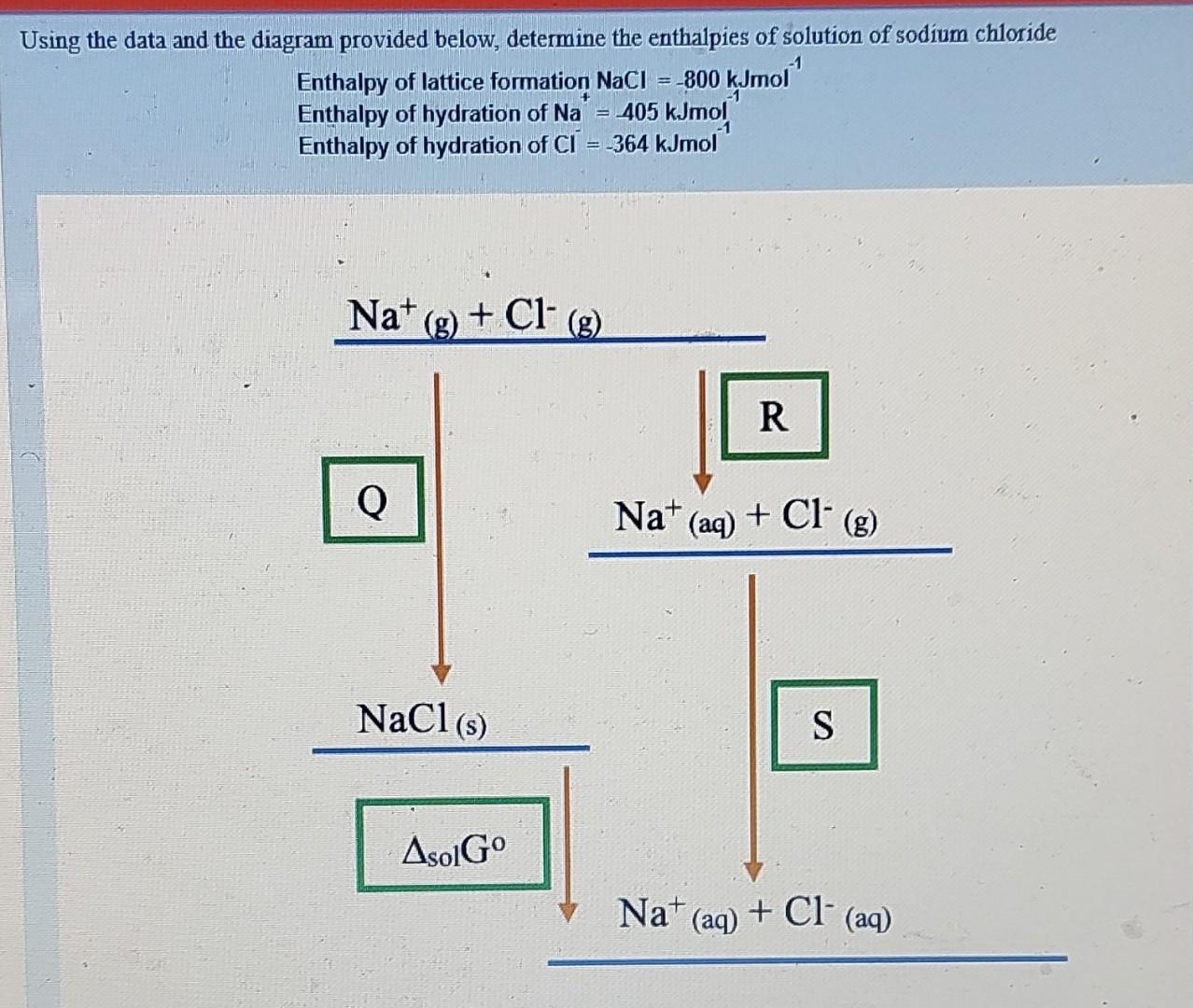
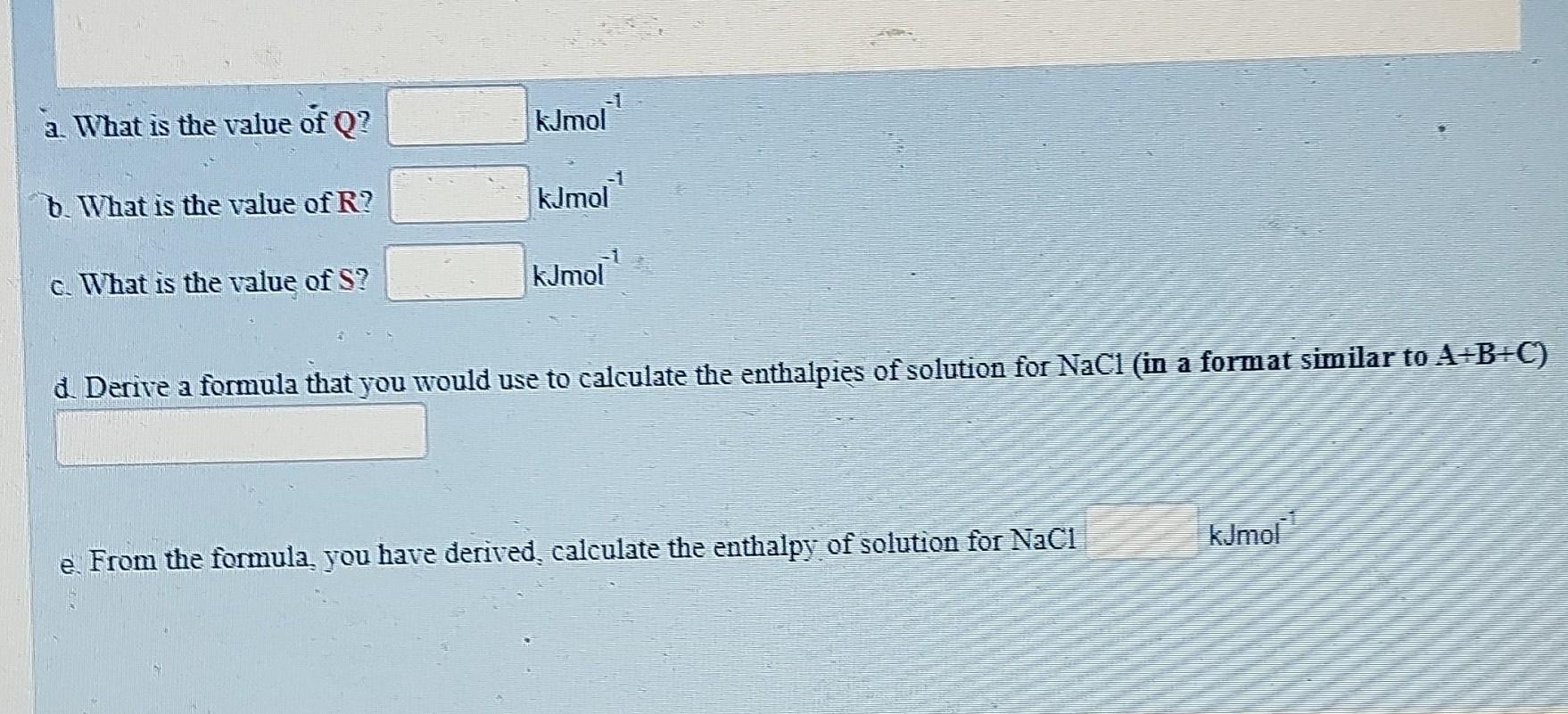
Using the data and the diagram provided below, determine the enthalpies of solution of sodium chloride Enthalpy of lattice formation NaCl=800kJmol1 Enthalpy of hydration of Na+=405kJmol1 Enthalpy of hydration of CI=364kJmol1 a. What is the value of Q ? kJmol 1 b. What is the value of R ? kJmol1 c. What is the value of S ? kJmol1 a. Derive a formula that you would use to calculate the enthalpies of solution for NaCl (in a format similar to A+B+C ) e. From the formula, you have derived, calculate the enthalpy of solution for NaCl kJmol1
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


