Answered step by step
Verified Expert Solution
Question
1 Approved Answer
What volume of 0.150MHClO4 solution is needed to neutralize 46.00mL of 0.0880MNaOH ? Express the volume in milliliters to two significant figures. Part B What
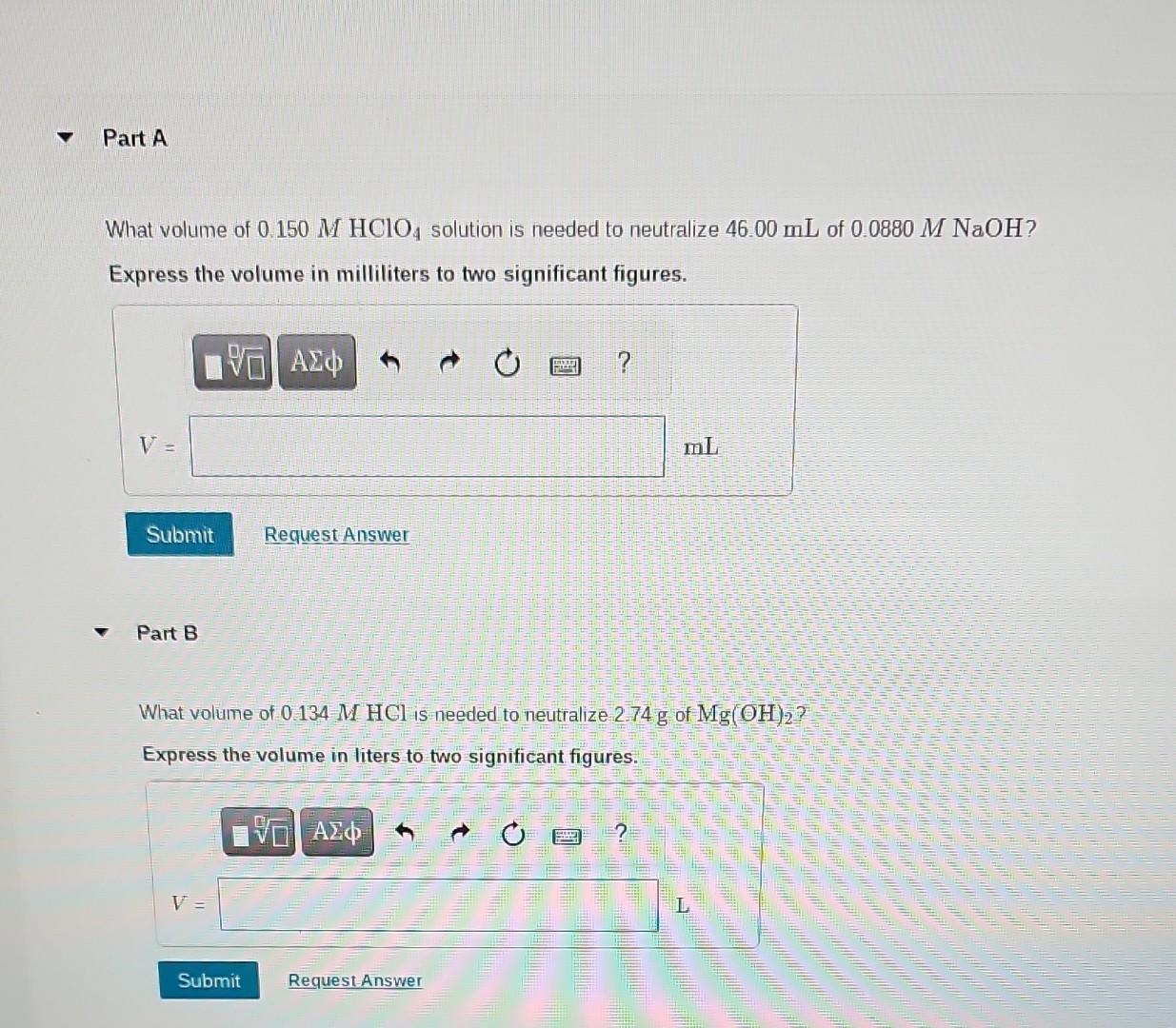
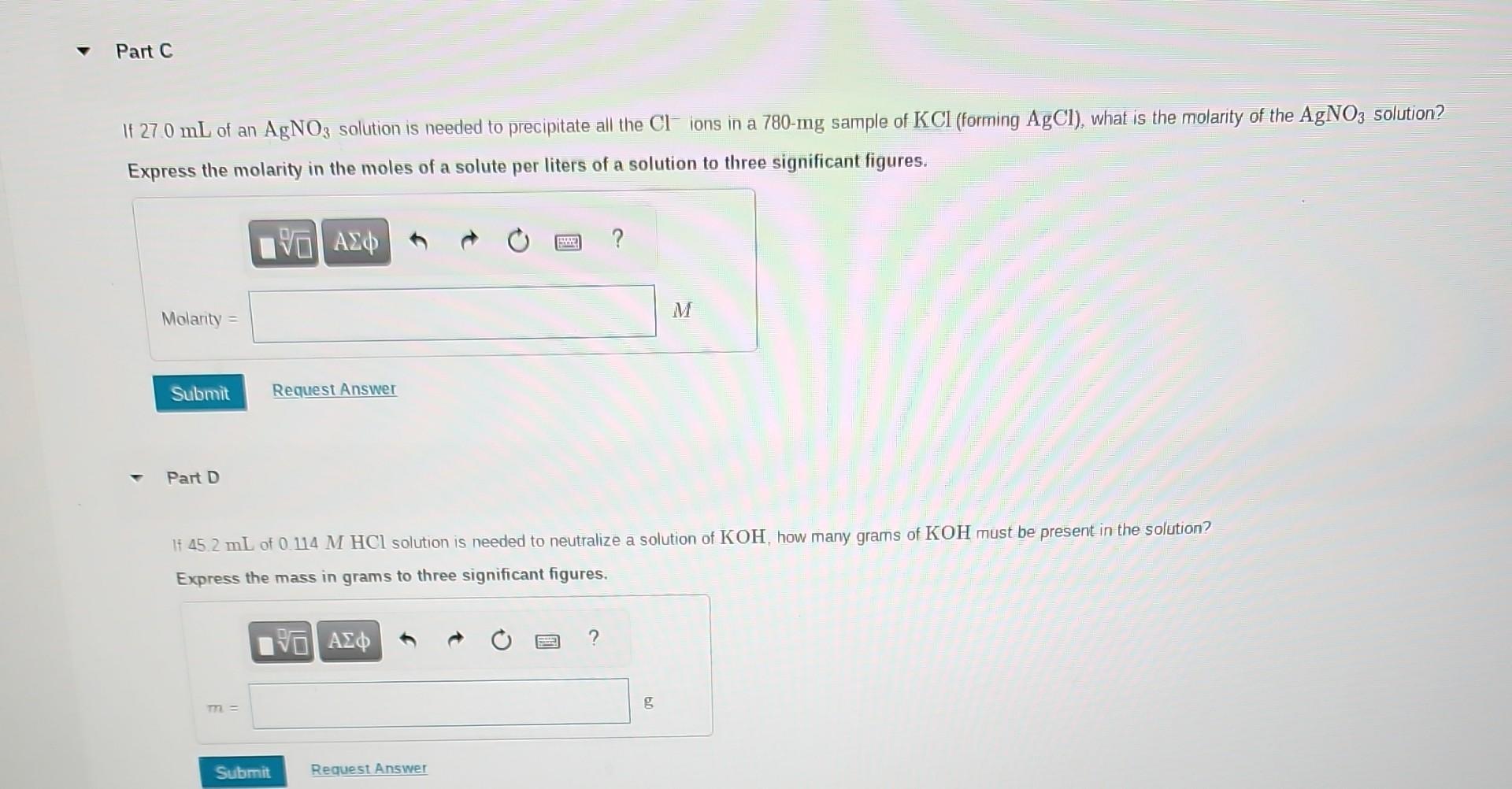
What volume of 0.150MHClO4 solution is needed to neutralize 46.00mL of 0.0880MNaOH ? Express the volume in milliliters to two significant figures. Part B What volume of 0.134MHCl is needed to neutralize 2.74g of Mg(OH)2 ? Express the volume in liters to two significant figures. If 27.0mL of an AgNO3 solution is needed to precipitate all the Clions in a 780mg sample of KCl (forming AgCl ), what is the molarity of the AgNO solution? Express the molarity in the moles of a solute per liters of a solution to three significant figures. Part D If 45.2mL of 0.114MHCl solution is needed to neutralize a solution of KOH, how many grams of KOH must be present in the solution? Express the mass in grams to three significant figures
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


