Answered step by step
Verified Expert Solution
Question
1 Approved Answer
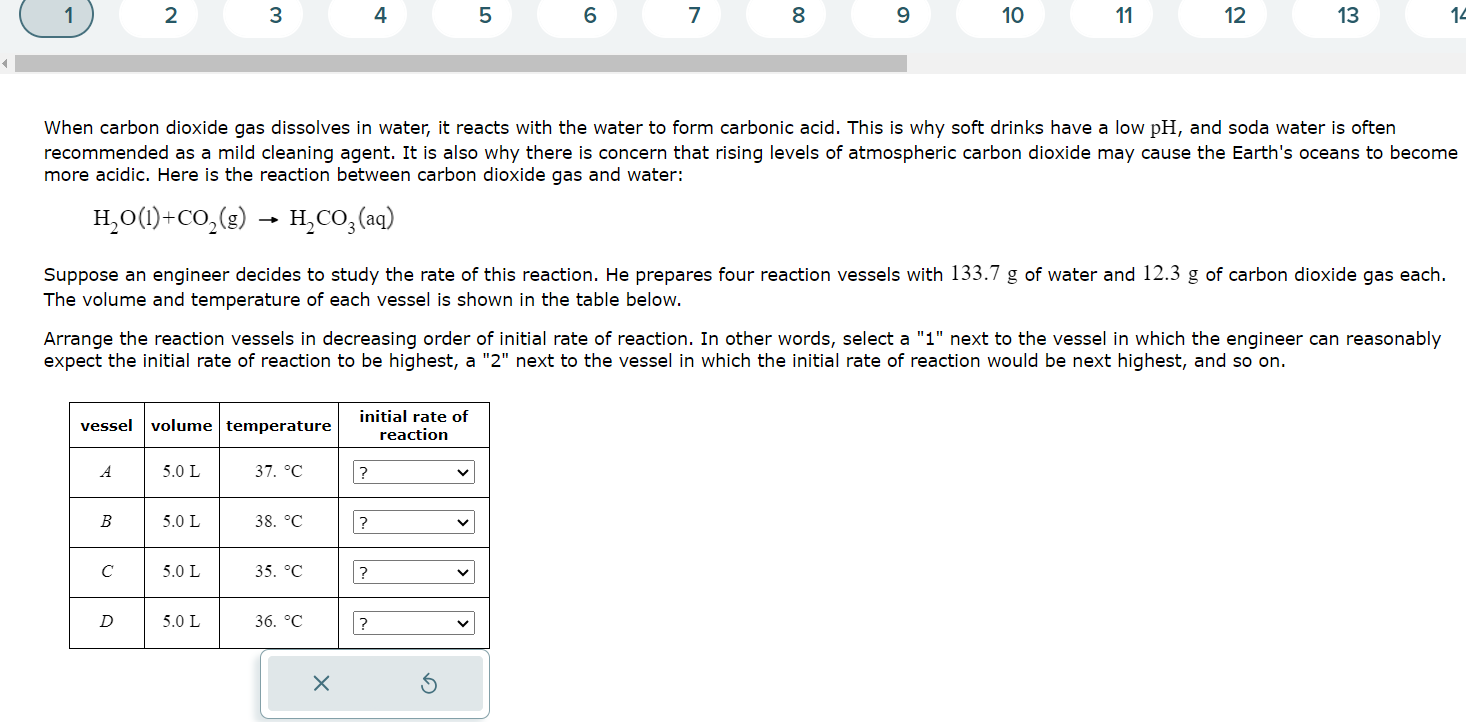
When carbon dioxide gas dissolves in water, it reacts with the water to form carbonic acid. This is why soft drinks have a low p
When carbon dioxide gas dissolves in water, it reacts with the water to form carbonic acid. This is why soft drinks have a low and soda water is often
recommended as a mild cleaning agent. It is also why there is concern that rising levels of atmospheric carbon dioxide may cause the Earth's oceans to become
more acidic. Here is the reaction between carbon dioxide gas and water:
Suppose an engineer decides to study the rate of this reaction. He prepares four reaction vessels with of water and of carbon dioxide gas each.
The volume and temperature of each vessel is shown in the table below.
Arrange the reaction vessels in decreasing order of initial rate of reaction. In other words, select a next to the vessel in which the engineer can reasonably
expect the initial rate of reaction to be highest, a next to the vessel in which the initial rate of reaction would be next highest, and so on
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


