Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Why is catalyst used in a chemical reaction? One of them is right answer: a)Because it increases the activation energy so that the reaction happens
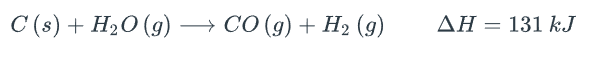
Why is catalyst used in a chemical reaction?
One of them is right answer:
a)Because it increases the activation energy so that the reaction happens faster
b)Because it lowers the activation energy so that the reaction takes place more slowly
c)Because the reaction then always becomes exothermic and thus happens faster
d)Because it lowers the activation energy so that the reaction happens faster.
C(s) + H2O(g) + CO(g) + H2 (9) AH = 131 kJStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


