Answered step by step
Verified Expert Solution
Question
1 Approved Answer
will upvote!! thanks! Problem 2 The reaction of ethyl-tetrabromide with zinc dust proceeds as shown in the flow chart below. Based on one pass through
will upvote!! thanks! 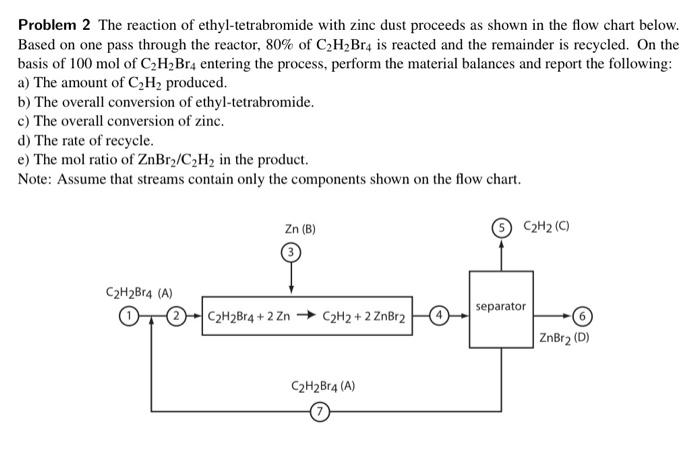
Problem 2 The reaction of ethyl-tetrabromide with zinc dust proceeds as shown in the flow chart below. Based on one pass through the reactor, 80% of C2H2Br4 is reacted and the remainder is recycled. On the basis of 100 mol of C2H_Br4 entering the process, perform the material balances and report the following: a) The amount of C2H2 produced. b) The overall conversion of ethyl-tetrabromide. c) The overall conversion of zinc. d) The rate of recycle. e) The mol ratio of ZnBry/C2H2 in the product. Note: Assume that streams contain only the components shown on the flow chart. Zn (B) C2H2 (0) C2H2B14 (A) separator C2H2Br4+2 Zn C2H2 + 2 ZnBr2 ZnBr2 (D) C2H2Br4 (A) Problem 2 The reaction of ethyl-tetrabromide with zinc dust proceeds as shown in the flow chart below. Based on one pass through the reactor, 80% of C2H2Br4 is reacted and the remainder is recycled. On the basis of 100 mol of C2H_Br4 entering the process, perform the material balances and report the following: a) The amount of C2H2 produced. b) The overall conversion of ethyl-tetrabromide. c) The overall conversion of zinc. d) The rate of recycle. e) The mol ratio of ZnBry/C2H2 in the product. Note: Assume that streams contain only the components shown on the flow chart. Zn (B) C2H2 (0) C2H2B14 (A) separator C2H2Br4+2 Zn C2H2 + 2 ZnBr2 ZnBr2 (D) C2H2Br4 (A) 
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


